Biology Quiz: Which organelle customizes proteins into forms the cell can use?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
11
views
pH
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#pH #HydrogenIons #HydroxideIons
SCIENCE ANIMATION TRANSCRIPT: Now that we've discussed acids and bases, let's talk about pH. The H in pH stands for hydrogen. pH is a measure of the concentration of free hydrogen ions in a solution. It tells us whether the solute in a solution is an acid or a base. Sometimes a base is referred to as being alkaline. To measure how acidic or basic a substance is, scientists use the pH scale, numbered from 1 to 14. The pH scale is an inverse scale of the concentration of hydrogen ions in a solution. The more hydrogen ions it contains, the lower its pH number and the stronger an acid it is. In contrast, the more hydroxide ions a solution has, the higher its pH number and the more basic or alkaline it is. Water is pH-neutral because it contains equal numbers of hydrogen and hydroxide ions. So water is right in the middle of the scale at a neutral pH of seven. Each whole number on the pH scale represents an exponential difference in value. This means that each whole number decrease in pH represents a 10 times increase in hydrogen ion concentration. For example, coffee has a pH of 5. So from a pH of 7 to a pH of 5, there is 10x10 or a 100-fold increase in hydrogen ion concentration compared to neutral water. As you can see, coffee is 100 times more acidic than water. This also means that coffee is 100 times less alkaline than water because it has a 100-fold decrease in hydroxide ion concentration. Baking soda is an example of a base with a pH of nine. It has a 100-fold decrease in hydrogen ion concentration compared to neutral water. However, it also has a 100 fold increase in hydroxide ion concentration. So baking soda is a hundred times more basic or alkaline than water. To function properly, biological processes in the human body require fluids with a pH in the range of approximately 6.5 to 7.5. For example, the normal pH of blood is 7.4. If acids or bases in the blood lower or raise the pH too much, a person could become sick or die. One way the body controls pH is through the use of different buffers. Buffers are substances that neutralize other acids and bases in solutions such as blood to prevent dramatic swings in pH. In summary, pH is a measure of the concentration of free hydrogen ions in a solution. A pH scale is an inverse scale of hydrogen ion concentration from 1 to 14. If the hydrogen ion and hydroxide ion concentrations are the same, the solution is pH-neutral. A pH of seven on the pH scale is neutral. Pure water has a pH of seven. Acids have a pH of less than seven and bases have a pH of greater than seven. The closer to seven a solution is, the weaker the acid or base. The farther away from seven a solution is, the stronger the acid or base. A buffer is a weak acid or base that reacts with a strong acid or base to prevent large changes in pH.
NSV16024
61
views
Biology Quiz: What sac-like organelle stores material within the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
2
views
Acids and Bases
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#acids #bases #IonicCompounds
SCIENCE ANIMATION TRANSCRIPT: What are acids and bases? First, recall that water is a polar molecule that dissolves ionic compounds by separating them into negatively-and positively charged ions. An acid is an example of one of these ionic compounds. A sample of this type of substance contains an abundance of positively-charged hydrogen ions that are released from the compound when dissolved in water. A base is also an ionic compound but a sample of this type of substance contains an abundance of negatively-charged hydroxide ions that are released from the compound when dissolved in water. An acid always has a greater concentration of hydrogen ions than hydroxide ions. In contrast, a base always has a greater concentration of hydroxide ions than hydrogen ions. If hydrogen ions and hydroxide ions are present in equal numbers, then the substance is neither an acid nor a base but is neutral like pure water. In summary, an acid is an ionic compound that releases many hydrogen ions when dissolved in water. A base is an ionic compound that releases many hydroxide ions when dissolved in water. Acids have a greater concentration of hydrogen ions than hydroxide ions. Bases have a greater concentration of hydroxide ions than hydrogen ions and neutral substances have equal numbers of hydrogen ions and hydroxide ions.
NSV16022
14
views
Mixtures with Water
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#mixtures #HeterogeneousMixture #HomogeneousMixture
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to talk about mixtures, specifically mixtures with water. A mixture is a physical, not a chemical, combination of two or more substances. Each substance in a mixture keeps its individual characteristics. A solution, also called a homogeneous mixture, consists of very tiny particles mixed so uniformly that the mixture has the same properties throughout. In contrast, a heterogeneous mixture consists of significantly larger particles that are not uniformly distributed and are more easily seen. The components of a heterogeneous mixture can usually be separated easily. Homogeneous water-based mixtures are called aqueous solutions. The dissolved substance is called the solute, and the substance that dissolves the solute, in this case water, is called the solvent. Water is sometimes referred to as the universal solvent because it can dissolve more substances than any other liquid. Water can dissolve many substances because of the polar nature of water molecules. This allows water molecules to surround and hold on to other small polar molecules. As a result, the polar molecules spread out evenly throughout the water. Remember, water is the solvent. Water's polar nature also allows the slightly positive and slightly negative charges on its molecules to dissolve ionic compounds known as salts. Water does this by separating the compound into negatively and positively charged particles called ions. The negative poles of water molecules surround the positive ion from the compound, and the positive poles of water molecules surround the negative ion from the compound. Solutes and solutions are so small and uniformly distributed that solutions are always transparent. You can see right through them. Now, let's talk about heterogeneous aqueous mixtures. There are two main types: colloids and suspensions. Colloids and suspensions differ primarily in the size of their particles within the water. In colloids, the particles mixed in the water are larger than the water molecules, but are still too small to see with the naked eye. As big as these particles are, they're still small enough that the random motion of the water molecules keep them mixed within the water. In contrast, suspensions contain even larger particles than those in colloids, but the particles are just small enough to be suspended in water when stirred or shaken. Over time, however, the particles in a suspension start to settle to the bottom of the container. Heterogeneous water mixtures such as colloids and suspensions are never transparent. They're always cloudy, hazy, or opaque, and you can't see through them. In this example, the colloid is milk, and the suspension is sand in water. To recap, mixtures are physical rather than chemical combinations of two or more substances, and each substance in the mixture keeps its individual characteristics. A mixture may be a solution which is a homogeneous mixture consisting of very tiny particles mixed so uniformly that the mixture has the same properties throughout, or a mixture may be heterogeneous with larger particles that are not uniformly distributed and are more easily seen. Aqueous solutions are homogeneous water-based mixtures consisting of tiny ions or molecules dissolved in water. A solute is the dissolved substance in a solution. A solvent is the substance that dissolves the solute. Water is called the universal solvent because it dissolves more substances than any other liquid. Aqueous colloids and suspensions are heterogeneous mixtures consisting of larger particles that are less uniformly distributed and more easily separated than the particles in aqueous solutions. [music]
NSV16028
24
views
Chemical Reactions
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#ChemicalReactions #ChemicalBonds #ChemicalEquation
SCIENCE ANIMATION TRANSCRIPT: What are chemical reactions? A chemical reaction is a process in which substances interact to form different substances by breaking, forming, or rearranging their chemical bonds. Substances that take part in a chemical reaction may be ionic or covalent compounds, as well as the atoms, ions, or molecules of some elements. Let's look at an example of a chemical reaction. Photosynthesis is a chemical reaction in which plants make food by using the sun's energy to combine carbon dioxide from the air and water from the soil into a sugar called glucose and oxygen. In this reaction, carbon dioxide and water are called reactants, because they're the substances changing by combining or reacting together. Glucose and oxygen are called products because they're the new substances produced by the reaction. Scientists write a chemical reaction in the form of a chemical equation. A chemical equation includes the reactants' chemical formulas on the left and the products' chemical formulas on the right. Notice that the reactants and the products are built from the same elements, carbon, oxygen, and hydrogen. Products must contain the same amount and type of elements that were in the reactants, and products never contain different elements than the reactants. In this reaction, the chemical formulas for the reactants are CO2 for carbon dioxide, and H2O for water. The chemical formulas for the products are C6H12O6 for glucose, and O2 for oxygen. We are not done yet because the equation is not balanced. This is necessary due to the law of conservation of matter, it states that matter can't be created or destroyed, but can only change forms. So to balance the equation, we have to make sure that the number of atoms of each element in the reactants is equal to those in the products, this is because a chemical reaction must abide by the law of conservation of matter, no atoms are created or destroyed. Here's the balanced version of this equation. It shows six molecules of carbon dioxide reacting with six molecules of water to produce one molecule of glucose and six molecules of oxygen. The numbers in front of the formulas are called coefficients. They indicate the number of molecules of each reactant and product. If there's no coefficient, it's understood to mean one molecule of that substance. To review, in a chemical reaction chemical bonds in reacting substances are broken then reformed to make different substances. The reacting substances are called reactants. The substances produced are called products. A chemical equation is a written expression of a chemical reaction, it includes the chemical formulas of both the reactants and the products. In a balanced chemical equation, the number of atoms of each element in the reactants equals those in the products. [music]
NSV16026
25
views
Van der Waals Forces
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#VanDerWaals #molecules #MolecularAttraction
SCIENCE ANIMATION TRANSCRIPT: In this video, we'll discuss Van der Waals forces. Van der Waals forces are forces of attraction between molecules that are very close together. These forces between molecules are much weaker than the chemical bonds between the atoms holding a molecule together. Let's see how Van der Waals forces work. Molecules are electrically neutral because they have equal numbers of positively charged protons in the nucleus and negatively charged electrons outside the nucleus. In addition, some molecules are also polar. What does this mean? Well, polar molecules have permanent poles of electrical charge like a magnet because the electrons are unevenly distributed around the molecule. How does this happen? Let's look at an example of a polar molecule, water. A water molecule, or H2O, consists of two hydrogen atoms and one oxygen atom. When a water molecule forms, both hydrogen atoms bond with the oxygen atom by sharing their electrons with the oxygen atom. This completes both oxygen's outer electron shell, which can hold all eight electrons, and hydrogen's outer shell, which can hold two. However, the electrons aren't shared equally between the atoms because the oxygen atom attracts the electrons more strongly than hydrogen. As a result, a partial negative charge develops around oxygen because there are more negatively charged electrons around the oxygen side of the molecule. In comparison, fewer electrons around the hydrogen atoms create a partial positive charge on the hydrogen side of the molecule. This unequal sharing of electrons creates opposing poles of electrical charge on either side of the two bonds that hold the atoms together. Because of the opposite poles, these bonds are called polar covalent bonds. And since a water molecule is angled or bent with both of the hydrogen atoms on one side and the oxygen atoms on the other side, the molecule as a whole also has opposite poles and therefore is referred to as a polar molecule. Now, when polar molecules are near each other, a Van der Waals force of attraction between the molecules occurs because of their oppositely charged poles. In this example, the attraction of a polar molecule's negative pole to the positive pole around hydrogen atoms in water is a particularly strong type of Van der Waals force called a hydrogen bond. Hydrogen bonds only occur in polar molecules between hydrogen in one molecule and oxygen, nitrogen, and fluorine in the other. If a molecule doesn't have permanent poles of opposite electrical charge, it's called a non-polar molecule. However, non-polar molecules can become polar for very brief moments since the locations of electrons around atoms are constantly changing. This means the molecule can have a temporary negative pole on the side where there are momentarily more electrons, and a temporary positive pole on the opposite side where there are fewer electrons. The momentary concentration of electrons in this molecule's negative pole can repel the electrons in a nearby molecule toward its opposite end, making the neighboring molecule polar as well. The oppositely charged poles of adjacent molecules attract each other, forming weak connections between them called Van der Waals forces. Van der Waals forces explains two important properties: cohesion, the attraction between like molecules within a substance, and adhesion, the attraction between unlike molecules in different substances. An example of cohesion is when opposite poles of water molecules are attracted to each other but not to the surrounding air. This creates an inward force allowing water to bead up and form water droplets. Adhesion, the force of attraction between unlike molecules, explains how geckos are able to climb on slick, flat surfaces. Although each molecular connection is very weak, geckos can form millions of them between the molecules within the microscopic hairs on each foot and the molecules in the climbing surface. These connections add up to more than enough adhesion force to support the gecko's weight. In summary, Van der Waals forces are forces of attraction between molecules. They are not the same as chemical bonds between atoms within a molecule. They can occur in permanently polar molecules, such as water, and in non-polar molecules when they become briefly polar due to the changing positions of electrons. A hydrogen bond is a strong Van der Waals force between a polar molecule containing hydrogen atoms and the negative pole of another polar molecule. Van der Waals forces account for cohesion, the attraction between like molecules within a substance, and adhesion, the attraction between unlike molecules in different substances. [music]
NSV16025
57
views
Cirugía bariátrica robótica
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Si usted sufre de obesidad severa y no ha podido perder peso, su médico puede recomendarle una cirugía bariátrica robótica. Su sistema digestivo o tracto gastrointestinal incluye: boca, esófago, estómago, intestino delgado e intestino grueso. Su páncreas, hígado y vesícula biliar libera jugos digestivos en su intestino delgado para ayudar a descomponer el alimento que usted come. Su médico puede recomendar cirugía bariátrica si: no puede perder peso y mantiene el peso a pesar de dieta y ejercicios, tiene un índice de masa corporal o IMC por encima de 40, tiene un IMC por encima de 35 y tiene una condición que amenaza su vida tal como diabetes o enfermedad cardíaca o pesa más de 100 libras, 45 kilos por encima de su peso corporal ideal o IBW. La cirugía bariátrica lo ayuda a perder peso, disminuyendo el tamaño de su estómago para que conserve menos alimento y se sienta satisfecho más rápidamente. Su procedimiento puede incluir un bypass en alguna parte de su intestino delgado para disminuir la longitud del camino del alimento a través del sistema digestivo así como la cantidad de calorías absorbidas del alimento. Antes del procedimiento, se colocará una vía intravenosa. Usted puede recibir antibióticos vía intravenosa para disminuir la posibilidad de infecciones. Se le administrará anestesia general. Se le insertará un tubo respiratorio por la boca y en la tráquea para ayudarle a respirar durante la operación. Su cirujano realizará una pequeña incisión cerca de su ombligo e insertará un tubo plástico llamado puerto. Se bombeará gas, dióxido de carbono, a su abdomen a través de este puerto. El gas inflará su abdomen lo que le dará al cirujano más espacio para ver y mover las herramientas quirúrgicas. Después de inflar su abdomen, se insertará una cámara de alta definición en este puerto. Su cirujano realizará incisiones adicionales del puerto para instrumentos robóticos así como par instrumentos usados por los asistentes que se encuentran junto al paciente. Un asistente insertará todas las herramientas robóticas por estos puertos. A diferencia de los instrumentos laparoscópicos estándar, estas herramientas pueden rotar 360° y tienen mayor flexibilidad que la muñeca humana. Sentado en una consola especial, su cirujano operará los brazos robóticos y la cámara con controles tipo joystick y pedales a pie. Una computadora traducirá los movimientos exactos de los dedos de su cirujano en movimientos preciosos de las herramientas quirúrgicas. A la vez, un sistema de visión de alta definición ofrecerá una vista estereoscópica aumentada en tres dimensiones del área quirúrgica. Dos procedimientos quirúrgicos bariátricos robóticos comunes son: el bypass gástrico y la banda gástrica ajustable. Si usted va a realizarse un procedimiento de banda gástrica ajustable, su cirujano aplicará una banda restrictiva alrededor de su estómago para limitar la cantidad de alimento que su estómago puede recibir. Un tubo conectará la banda a un puerto justo debajo de la piel de su abdomen. Su cirujano inyectará una solución salina en el tubo para ajustar la rigidez de la banda, según sea necesario, para acelerar la pérdida de peso o reducir los efectos colaterales. Si usted va a realizarse un procedimiento de bypass gástrico, su cirujano creará una pequeña bolsa en su estómago y la separará del resto de su estómago y la parte superior de su intestino delgado. La sección inferior de su intestino delgado se unirá a la bolsa del estómago. Luego, su cirujano volverá a unir la sección superior de su intestino delgado a una parte diferente de su intestino delgado inferior, lo que permitirá que los jugos digestivos de su páncreas, hígado y vesícula biliar ayuden a digerir los alimentos. Al final de cualquier de los dos procedimientos, se cerrarán las pequeñas incisiones con puntos de sutura, grapas, pegamento quirúrgico o con vendajes de cierre. Después del procedimiento se le retirará el tubo respiratorio y se le trasladará al área de recuperación para controlarlo. Le darán medicamentos para el dolor si fuese necesario. Usted puede continuar recibiendo antibióticos por vía intravenosa. La mayoría de los pacientes son dados de alta del hospital uno o dos días después del procedimiento.
ANR12005es
20
views
1
comment
Ionic vs. Covalent Bonds
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#ChemicalBonds #IonicBonds #CovalentBonds
SCIENCE ANIMATION TRANSCRIPT: In this video, we will compare ionic and covalent bonds. In order to understand ionic bonds, we need to talk about ions first. Atoms are electrically neutral because they have equal numbers of both positively charged protons and negatively charged electrons. However, an atom can become a charged particle called an ion if it gains or loses electrons. If an atom gains electrons, it acquires more negative charge. As a result, it becomes a negatively charged ion. Conversely, if an atom loses electrons, it loses some of its negative charge and becomes a positively charged ion. The only way to get a positively charged ion is to lose negatively charged electrons. Remember, you can't just add a proton to make a positive ion because changing the number of protons would change it into a different element. Now, let's talk about ionic bonds. Notice that this term contains the word ion. That's because ionic bonds create ions out of electrically neutral atoms by the transfer of one or more valence electrons from one atom to another. Further, electrically neutral atoms of elements whose outer shell is less than half filled with valence electrons tend to donate electrons, while atoms whose outer shell is more than half filled tend to attract electrons. For example, sodium and chlorine atoms are electrically neutral. Chlorine, which only needs one electron to fill its outer shell, strongly attracts sodium's single valence electron. So, these elements react to form a chemical bond, creating sodium chloride. Sodium chloride, otherwise known as table salt, is an example of an ionically bonded compound. This is because the electrically neutral sodium atom became a positively charged ion by losing its valence electron. And chlorine became a negatively charged ion by gaining this electron from sodium. So, how do covalent bonds occur? The simplest substance that contains a covalent bond is a molecule of hydrogen gas also known as H2. A hydrogen atom has only one electron in its outer shell, which for this atom is also the shell nearest the nucleus. This shell can hold a maximum of two electrons. So, atoms of hydrogen tend to pair up and share their electrons so that both atoms have their outer shell filled. As you can see, covalent bonds occur when atoms share pairs of electrons. In this molecule, the hydrogen atoms form a single covalent bond. Another example of a covalently bonded molecule is carbon dioxide, or CO2. From its chemical formula, you know that carbon dioxide contains one carbon atom and two oxygen atoms. Carbon has four valence electrons, and both oxygen atoms have six valence electrons. But all three atoms would need eight electrons to fill their outer shells. So, each oxygen atom shares a pair of electrons with the pair of electrons in carbon. This results in two double covalent bonds where two pairs of electrons are shared between each atom. To summarize, the two main types of chemical bonds are ionic bonds and covalent bonds. In ionic bonds, one or more electrons are transferred from one atom to another. In covalent bonds, one or more pairs of electrons are shared between atoms. [music]
NSV16029
170
views
Overview of Chemical Bonds
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#ChemicalBonds #IonicBonds #CovalentBonds
SCIENCE ANIMATION TRANSCRIPT: This video is an overview of chemical bonds. How do the atoms of elements form chemical bonds? Recall that electrons and an atom surround the nucleus. They feel energy levels or shells in specific numbers. Electrons in an atom's inner shells are commonly referred to as core electrons. Core electrons don't participate in chemical bonds. In contrast, electrons in the outermost shell of an atom are called valence electrons. Valence electrons do participate in forming chemical bonds. For example, a carbon atom with six protons and six neutrons in the nucleus has six electrons. Notice that carbon has both core and valence electrons. The innermost shell of any atom can hold a maximum of two electrons and the next shell can hold up to eight electrons. So, two of the electrons in carbon fill the first shell, and the remaining four electrons are in the next shell. The two electrons in the first shell are carbon's core electrons. The four electrons in the outer shell are carbon's valence electrons. Atoms with fewer valence electrons than its outer shell can hold aren't as stable as atoms with full outer shells. However, these atoms can become more stable if their outer shell is filled. This can happen either by loosing electrons to another atom or attracting electrons from another atom. This interaction of valence electrons between atoms results in the formation of chemical bonds. Elements that have completely filled outer shells such as helium are mostly non-reactive which means, they don't usually form chemical bonds. Why is that? Well, it's because their outer shells can't accept anymore electrons and losing all of their valence electrons requires too much energy. There are two main types of chemical bonds, they are ionic bonds, when electrons are transferred from one atom to another, and covalent bonds, when atoms share electrons. We'll discuss this in more detail separately. [music]
NSV16020
9
views
Chemical Compounds
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#ChemicalCompounds #ChemicalFormula #AtomicBonds
SCIENCE ANIMATION TRANSCRIPT: Now that you've learned about atomic structure and elements, let's look at chemical compounds. A chemical compound is a substance made of two or more elements that are chemically bonded together in fixed proportions. Common examples are water and table salt. You've probably heard of H2O. That's a chemical formula for water. In the chemical formula for water, the little number slightly below the letter H for hydrogen is called a subscript. A subscript in the chemical formula tells you how many atoms of that element are in one unit of the compound. There is no subscript next to the O for oxygen. That means there's only one oxygen atom. We don't write the one. When there is no subscript it's understood that we mean one atom. So, H2O is the formula for one unit of water. The formula shows that a unit of water contains a fixed proportion of two atoms of hydrogen to one atom of oxygen. It's important to know that compounds usually have very different physical and chemical properties than the individual elements they contain. For example, although water is liquid at room temperature, the elements hydrogen and oxygen are gasses. The next compound we'll look at is sodium chloride, commonly known as table salt. Sodium chloride consists of sodium and chloride ions. Ions are charged particles because they have gained or lost electrons. How many ions of sodium are there in one unit of the chemical formula for sodium chloride? No subscript means there's only one sodium ion. And how many chloride ions? Again, there's only one. So, sodium chloride has a one-to-one ratio of sodium to chloride ions. Okay. The last example of a compound is glucose. A simple sugar your body cells use for energy. The chemical formula for one unit of glucose is C6H12O6. Here we can see that one unit of glucose has six carbon atoms and 12 hydrogen atoms. How many oxygen atoms does one unit of glucose have? If you said six oxygen atoms, you'd be right. In review, a compound is a substance containing the chemically bonded atoms of two or more elements. A chemical formula gives the proportional number of atoms or ions of each element in a compound. And the compound usually has different physical and chemical properties than the elements it contains. We'll explore how elements chemically bond together in the next video. [music]
NSV15015
123
views
Cáncer de ovario
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Los ovarios son las glándulas reproductivas femeninas. Se componen de tres tipos de células: epitelial, germinal y estromal. Cuando las células en los ovarios crecen de forma anormal, se convierten en tumores que pueden ser benignos o cancerosos. Si los tumores cancerosos no se detectan a tiempo, las células cancerosas pueden propagarse desde los ovarios, a través de la región pélvica y continuar propagándose hacia la zona abdominal y otros órganos. La determinación de la etapa del cáncer de ovario es muy importante en el desarrollo de un plan de tratamiento. En la etapa uno, el cáncer se limita a uno o ambos ovarios. En la etapa dos, el cáncer de ovario se extiende hacia la zona de la pelvis. En la etapa tres, el cáncer de ovario se ha propagado hacia la región abdominal. En la etapa cuatro, el cáncer de ovario se extiende hacia el hígado o ubicaciones fuera del área abdominal. Generalmente, el tratamiento del cáncer de ovario incluye cirugía, quimioterapia, terapia de radiación o una combinación de terapias.
ANH14136es
6
views
Periodic Table
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#PeriodicTable #Elements #AtomicNumber
SCIENCE ANIMATION TRANSCRIPT: In this video, we'll discuss the periodic table of elements. A chart called the periodic table of the elements organizes all of the known elements. Rows in the periodic table are called periods, and columns are called groups or families. A section from each of the two bottom periods has been pulled out and placed below the table to avoid making the table too wide. The elements are organized left to right and top to bottom by their atomic number, meaning the number of protons in one atom of the element. Each box shows an element represented by its unique symbol. The smaller number next to each element's symbol represents the atomic number. The atomic number increases by one as you go from left to right across each period. The larger number represents the atomic mass. Notice that the atomic mass of many elements is a decimal number rather than a whole number. This is because the atomic mass is a weighted average of the mass numbers for the isotopes of an element. A weighted average takes into account how common each isotope of an element is in nature. The most common isotope counts for much more in the average than less common isotopes, just like a final exam may count more than quizzes towards your grade in a class. Notice that the atomic mass isn't the same as mass number, which is simply the total number of protons and neutrons in the nucleus of a particular isotope. However, you can determine the mass number of an element's most common isotope by rounding its atomic mass up or down to the nearest whole number. You can do this because the most common isotope has the most influence on the atomic mass. Round up if an element's atomic mass ends in .5 or greater, round down if an element's atomic mass ends in less than .5. Let's look at some examples from the periodic table. Helium has an atomic mass of 4.003. We can easily round that down to get a mass number of 4. We can also see that helium's atomic number is 2, which means it has 2 protons. Now, we can subtract the atomic number from the mass number to see that the most common isotope of helium has 2 neutrons. In the case of oxygen, we can round its atomic mass up to get a mass number of 16. Since its atomic number is 8, we know oxygen has 8 protons. And by simple subtraction, we can determine oxygen also has 8 neutrons. How does this work in a less common isotope of an element, such as hydrogen-3? The most common hydrogen isotope is hydrogen-1, as you can see from rounding the atomic mass listed in the periodic table. Recall that isotopes are identified by their mass number. So, we know hydrogen-3's mass number is 3. So, we can subtract hydrogen's atomic number of 1 from its mass number and see that hydrogen-3 has 2 neutrons. In summary, the periodic table is an organization chart of all the known elements. Each element is represented by its symbol, atomic number, and atomic mass. Elements are arranged left to right and top to bottom by increasing atomic number. An element's atomic mass is a weighted average of its isotope's mass numbers. Round the element's atomic mass up or down to find the mass number of its most common isotope. [music]
NSV15014
45
views
Elements
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#elements #AtomicNumber #Chemistry
SCIENCE ANIMATION TRANSCRIPT: What is an element? Elements are pure substances that are made up of only one type of atom such as hydrogen, carbon, or mercury. So, what makes one element different from another? Well, it's the number of protons in a single atom of an element. This is called the element's atomic number. For example, hydrogen has one proton in its nucleus, so its atomic number is one. Carbon has six protons, so its atomic number is six. And mercury's atomic number is 80 because it has 80 protons in its nucleus. Since the nucleus contains almost the entire mass of an atom, the number of particles it contains has a big effect on the atom's mass. Each positively charged proton has a mass unit of one, and each neutral neutron also has a mass unit of one. The total number of protons and neutrons in the nucleus of an atom is called the mass number. In this example, the mass number of a hydrogen atom is one. Hydrogen is the only element that usually doesn't have any neutrons. The mass number of this carbon atom with six protons and six neutrons is 12. And the mass number of this mercury atom with 80 protons and 121 neutrons is 201. Needless to say, mercury is a match heavier element than hydrogen or carbon. Even though every atom of the same element always has the same number of protons, sometimes an element has atoms with different numbers of neutrons. Ordinary hydrogen has no neutrons, but there's a version of hydrogen with one neutron and another version with two neutrons. Atoms of the same element with different numbers of neutrons are called isotopes. The three isotopes you see here are all still hydrogen because they all have only one proton. Since neutrons have about the same mass as protons, isotopes of the same element have different mass numbers. In fact, an element's isotopes are often identified by their mass numbers. To sum up, an element is a pure substance made of atoms that always have the same number of protons. This means atoms with different numbers of protons are different elements. The number of protons in one atom of an element is called the atomic number. The number of protons plus neutrons in one atom is called the mass number. Isotopes are atoms of the same element with different numbers of neutrons. [music]
NSV16021
35
views
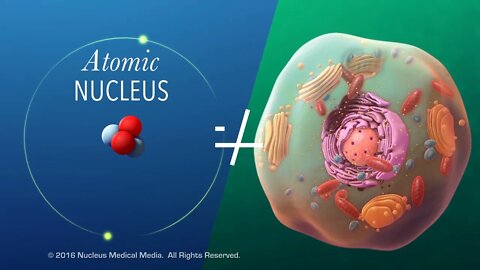
Atoms
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#atoms #chemistry #biology
SCIENCE ANIMATION TRANSCRIPT: In this video, we'll discuss what atoms are. The chemistry of life begins with understanding the properties of matter. Of course, matter is everything that has mass and takes up space. It includes both nonliving things as well as all living organisms. And atoms are the basic units of all matter, both living and nonliving. They're so small that you could fit billions of them on the head of a pin. But what are atoms made of? First, let's look at the inner central region called the nucleus. Nucleus means center. However, the atomic nucleus isn't the same thing as the nucleus in a cell. A cell's nucleus is its control center, containing instructions that control cell functions. An atomic nucleus is made up of one or more subatomic particles called protons and neutrons. If the nucleus has only one particle, it must be a proton. Each proton and neutron has an atomic mass unit of one. Together, these nuclear particles contain virtually the entire mass of an atom, but they take up less than 1% of the volume. In addition to mass, some subatomic particles also have an electrical charge. A proton carries a positive charge of plus one. As its name indicates, a neutron is electrically neutral, which means it has no charge. Outside the nucleus are subatomic particles called electrons. While electrons contribute almost nothing to the mass of an atom, each of them carries a negative electrical charge of minus one. And even though electrons are always outside the nucleus, they're found in layers called energy levels, or shells, around the nucleus. Each electron shell or energy level has a maximum number of electrons it can hold. For simplicity, diagrams of atoms often show electrons within these shells while orbiting the nucleus. However, since we can only know the probability of where they might be located, electrons are sometimes depicted as smeared out and fuzzy. This fuzzy view of electrons is called an electron cloud. Notice that neutral atoms contain equal numbers of protons and electrons. This means that the positive charge of the protons balances out the negative charge of the electrons, making the atom electrically neutral. In summary, an atom has three main subatomic particles: protons and neutrons in the nucleus, and electrons in shells outside the nucleus. The sum of the protons and neutrons in the nucleus makes up almost the entire mass of the atom. Protons have a positive electrical charge. Electrons have a negative electrical charge, and neutrons have no charge at all. Electrically neutral atoms have an equal number of protons and electrons. [music]
NSV15010
33
views
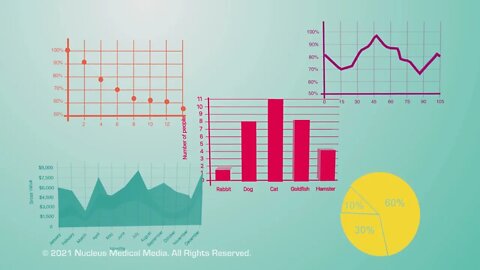
Biology 101: How to Understand Graphs
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#xyGraphs #LineGraphs #BarGraphs #AreaGraphs #PieCharts #biology
SCIENCE ANIMATION TRANSCRIPT: Let's look at different types of graphs and see how they work. Graphs are diagrams that display data in an organized and easy-to-read fashion. Note that all graphs must have a title that summarizes this data. There are different kinds of graphs, so it's important to know how to interpret each type. When you know how to do that, you will be able to create a graph of your own. The types of graphs we will look at are X-Y graphs, line graphs, area graphs, bar graphs, and pie charts. First, we'll look at X-Y graphs. X-Y graphs, also known as scatter plots, look at how two events or variables are possibly related. The horizontal X-axis shows data that represents the independent variable. Remember, the independent variable is the variable you're intentionally changing or testing in an experiment. For example, the independent variable might be how long a student studies. The vertical Y-axis shows data that represents the dependent variable. You may recall that the dependent variable is the outcome you're observing or measuring as a result of exposure to the independent variable. For example, the dependent variable could be the grade that corresponds with how long a student studied. You can use X-Y graphs to look for trends in the relationship between the independent and dependent variables. If the values of both of these variables rise, then a line connecting the data points will show an upward trend. This means the variables are positively correlated. Now, let's change the graph to show how class grades are affected as the study time goes down. If the values of both variables go down, then a line connecting the data points will show a downward trend on the graph. Note that this is also described as a positive correlation. A positive correlation means the values of both variables are increasing or that the values of both variables are decreasing. So, what is a negative correlation? In a negative correlation, one variable goes up while the other goes down. Here, we're showing how class grades might go down as the number of missed classes goes up. So a line connecting the data points will show a downward trend. Remember, variables are negatively correlated when one value is increasing while the other value is decreasing. In some cases, the variables might follow a random pattern and have no relationship. As an example, this graph plots student height with class grades. As you can see, these variables demonstrate no correlation. Now, let's talk about line graphs. Line graphs are used to track certain changes as measured on the Y-axis, usually, over a period of time, as measured on the X-axis. This line graph shows the number of magazines sold over the course of a week. Reading this graph, you can see the most magazines were sold on Thursday and the least number of magazines were sold on Friday. Next up are area graphs. Area graphs are a combination of multiple line graphs. When making an area graph, each line graph usually has a different color underneath, with a color key that identifies what each line represents. Area graphs are useful for comparing datasets and identifying trends, such as what items are hot sellers or weak sellers in each month. Now, we move to bar graphs. A bar graph can compare different groups, such as the number of people who own different pets. Like line graphs, a bar graph can also track changes over time. The last type of graph we'll talk about is a pie chart. A pie chart shows the various parts that make up a whole. A pie chart often looks like a pizza cut into uneven slices. Just like all the pizza slices put together make up 100% of the pizza pie, all the sections of a pie chart represent different amounts that add up to 100% of the total amount. For example, in a classroom of 30 students, this pie chart represents how many have brown eyes versus blue eyes versus green eyes. Pie charts don't show trends, they just show how things are distributed within a group. So to review, graphs are an organized way to show data. X-Y graphs show how an independent variable on the X-axis relates to a dependent variable on the Y-axis. Line graphs also have X and Y axes but track changes that take place usually over time. Area graphs are a combination of multiple line graphs. Bar graphs compare values or track changes over time. And pie charts show the various percentages of things within a whole group.
NSV16032
37
views
Qualitative and Quantitative Data
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#QualitativeData #QuantitativeData #biology
SCIENCE ANIMATION TRANSCRIPT: Let's look at the difference between qualitative and quantitative data. Scientists observe and collect different types of information called data. So, what kind of data can we collect? Well, the two main types of data are qualitative data and quantitative data. Qualitative data includes descriptions that do not contain numeric values. Notice the word qualitative has "quality" embedded in it. Qualitative data tends to be subjective impressions such as how tasty one school lunches compared to another one. As you can see, qualitative data consists of observable things that can be described and recorded in ways other than numerically. Now, let's talk about quantitative data. Unlike qualitative data, quantitative data contains numbers obtained by counting or measuring. Notice the word quantitative has "quantity" embedded in it. For example, recording quantitative data can be as simple as counting specific things such as the number of boys in your class compared to the number of girls. Quantitative data can also be measurements of length, width, height, volume, as well as mass, or temperature. Sometimes scientists will breakdown quantitative data into specific types called discrete data and continuous data. Discrete data can only have a certain exact value which can't be subdivided. For example, if you roll a typical pair of dice, you can roll a two or a three, but it's impossible to roll a two and a half. You can only roll a whole number between 2 and 12. Likewise, the number of protons in an atom is also an example of discrete data because you can't have half a proton. In contrast, continuous data can have almost any value. For example, measurements such as height come in a range of continuous data. Why? Because measurements can have any value in fractions of a unit, in this case, meters. In review, qualitative data contains descriptions that don't use numbers, while quantitative data contains numbers obtained by counting or measuring. Both discrete and continuous data are types of quantitative data. Discrete data can only contain certain specific values, while continuous data can have almost any value. [music]
NSV16031
7
views
Cómo controlar el Cáncer de Mama YT
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Usted o alguien que usted quiere puede haber sido diagnosticada con cáncer de mama. Este video le ayudará a entender cómo manejarlo. El cáncer de mama es un tipo de cáncer que comienza en las células de la mama. Para muchas mujeres, los tratamientos para el cáncer de mama pueden eliminar o destruir el cáncer. Después, usted debe recibir un plan de atención con seguimiento por parte de su equipo de atención médica. Esto incluye ir a las citas de seguimiento. Su médico querrá asegurarse de que el cáncer no ha vuelto y comprobar si hay problemas de salud resultantes del tratamiento. También es importante hacerse mamografías regulares para controlar el cáncer de mama según lo recomendado por su médico. Si usted está tomando algún medicamento, continúe tomándolo según los prescrito por su médico. Tome nota de cualquier nota de cualquier efecto secundario e infórmeselo a su médico. Si usted ha tenido cirugía de mama u otros tratamientos, siga las instrucciones que le dieron. También puede ser necesario hacer algunos de los siguientes cambios saludables a su estilo de vida. Coma una dieta saludable con más frutas, vegetales y granos enteros. Una dieta saludable puede reducir el riesgo de cáncer, o la posibilidad de que regrese. También es importante que usted permanezca físicamente activo. Actividades como caminar, andar en bicicleta o nadar pueden ayudarle a sentirse mejor y menos cansado. Pídale a su médico que le recomiende un fisioterapeuta o un especialista en ejercicio que pueda ayudarle a diseñar un plan de ejercicios adecuado para usted, y un nutricionista que lo ayuden a diseñar una dieta o plan de alimentación apropiado. Dejar de fumar y limitar el consumo de alcohol puede ayudar a reducir el riesgo de cáncer. Saber que tiene cáncer puede ser abrumador. Es posible que tenga preocupaciones acerca de su afección y cómo afecta a su familia, sus tratamientos y estancias en el hospital, gastos médicos, y su trabajo. Afortunadamente, hay maneras de lidiar con esto. Recuerde que su médico y el equipo de salud están allí para responder a cualquier pregunta que tenga. Las siguientes fuentes de apoyo pueden ayudarle a abordar sus preocupaciones: trabajadores sociales, líderes religiosos, consejeros y grupos de apoyo. Hable con su médico si tiene alguna pregunta sobre su plan de tratamiento, medicamentos o cambios de estilo de vida que la ayuden a manejar el cáncer de mama.
ANH15165es
10
views
Independent Variable vs Dependent Variable
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#IndependentVariable #DependentVariable #biology
SCIENCE ANIMATION TRANSCRIPT: Independent variable and dependent variable are important terms related to controlled experiments. Remember, a controlled experiment is a scientific test in which all conditions are kept constant except for the variable you're testing. The independent variable is the thing you're testing in an experiment. It's often abbreviated as IV, and sometimes called the manipulated variable because you change or manipulate this variable. In an experiment, the dependent variable is the thing you're observing and measuring, the thing you're anticipating may be affected as a result of exposure to the independent variable. It's often abbreviated as DV, and sometimes called the responding variable because it responds to the change that you make. Let's look at a few examples. Suppose your hypothesis is that if students study 15 minutes a night, then they will have higher test grades than those who don't study at all. What's being changed or manipulated? It's whether or not the students study for 15 minutes. And what's going to be observed or measured in this experiment? What do you think might be different as a result of this increased study time? You're expecting tests grades will be affected. In this experiment, the independent variable is the study time. And the dependent variable, the thing you're measuring or going to observe is the tests grades. Here's another example. See if you can figure it out. This time, the hypothesis is that if people who have headaches take aspirin, then they will get relief faster than those who don't take aspirin for headaches. So, what's the thing that's different in this case? The thing that's different, the independent variable, is whether or not somebody is taking an aspirin. Then what are you going to measure? You're measuring how long it takes for their headache to go away. That's the dependent variable. Here's a final example to help you understand these terms. You predict that if a brand name light bulb is left on continuously, then it will burn longer than a bargain brand light bulb used in the same manner. In this case, what is the independent variable? It's the brand name light bulb. What are you measuring? You're measuring how many hours the light bulbs work before burning out, which is the dependent variable. The independent variable is the brand name light bulb, and the dependent variable is the amount of time the light bulbs work before burning out. So, to review, the independent variable is the thing that you're testing. Sometimes this is referred to as the cost in an experiment. It is also the "if" part of your hypothesis. The dependent variable, the thing you're measuring, is the effect. It is also the "then" part of your hypothesis. [music]
NSV15011
8
views
Controlled Experiments
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#ControlledExperiment #ScientificHypothesis #biology
SCIENCE ANIMATION TRANSCRIPT: What is a controlled experiment, and why would you want to do one? Maybe you have an idea that you think might explain a situation. This is called a scientific hypothesis. How could you find out if your hypothesis is correct? Well, you'd set up a controlled experiment in which you control, or keep constant, all the factors, known as variables, except for the one you want to test. Let's design a controlled experiment to test a fertilizer which claims it makes plants grow bigger, lusher, and perhaps produce more flowers, fruit or vegetables. If you wanted to see if the fertilizer works, how would you set up a controlled experiment to test this claim? First, you would get two plants of the same species. Let's call them Plant A and Plant B. Everything about the plants should be exactly the same, including their size, health, and age. Next, you would put each plant in identical pots with the same amount of the same kind of dirt or soil. You would water them both the same amount at the same times. You would also put the plants next to each other in the same place, such a window sill, so that they're boh exposed to the same amount of sunlight and kept at the same temperature. It's important that everything is the same, because the purpose of your experiment is to find out whether or not the fertilizer works. So what would be different? In this experiment, the only difference is that only plant A would get the fertilizer. Now remember, your hypothesis is that Plant A, which is getting fertilizer, will grow bigger compared to Plant B, which isn't getting any fertilizer. How would you know whether your hypothesis is correct? You'd know because you'd regularly measure the plants during the course of the experiment, for example, once a week for a period of three months. You would record these measurements throughout the experiment. These measurements are your data. At the end of the experiment, you would look at your data and compare the measurements of Plant A, which got fertilizer, to Plant B, which didn't get fertilizer. As you can see, Plant A did grow bigger than Plant B. So, it appears that the results of this controlled experiment support your hypothesis. So, let's recap the elements of experimental design. What were you testing? You were testing to see whether or not fertilizer promotes plant growth. What was your hypothesis? The hypothesis was that the plant that got fertilizer would get bigger than the plant that didn't get fertilizer. What were you measuring? You measured the growth of both plants. How do you know if the results of the experiment support your hypothesis? If your hypothesis is true, you would have seen that the plant that got the fertilizer actually did get bigger than the plant that didn't get any fertilizer. The variable you were testing, in this case, the fertilizer, is called the independent variable. And the thing you were observing, measuring, and expecting to change because of that independent variable was plant growth. In this experiment, plant growth is the dependent variable. We'll go over independent and dependent variables in more detail in another video. [music]
NSV15012
23
views
Biology Quiz: What is the tangled, spread-out form of DNA?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
9
views
Biology Quiz: What condensed structures do DNA form when the cells is ready to divide?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
4
views
Biology Quiz: What structure in the nucleus makes ribosomes?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
7
views
Biology Quiz: What do Ribosomes do?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
11
views
Biology Quiz: What is the jelly-like substance inside the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
9
views