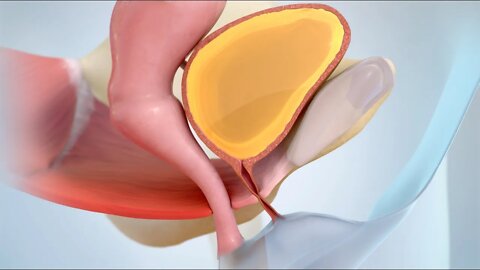
Cómo controlar el cáncer de ovario
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
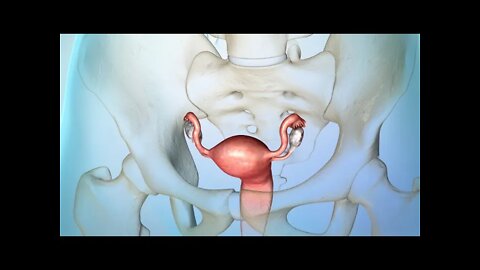
MEDICAL ANITION ANSCRIPT: Usted o alguien que usted quiere puede haber sido diagnosticada con cáncer de ovario. Este video le ayudará a entender cómo manejarlo. Los ovarios son dos glándulas que están en la pelvis de la mujer. Estos producen hormonas y óvulos para la reproducción. El cáncer de ovario es una enfermedad en la cual las células cancerosas crecen sin control en sus ovarios. El tratamiento para el cáncer de ovario puede eliminar o destruir el cáncer. . Después del tratamiento, es muy importante tener un plan de cuidado con seguimiento. Esto incluye ir a todas las citas de seguimiento. Su médico querrá asegurarse de que el cáncer no ha reaparecido además de comprobar si hay problemas de salud resultantes del tratamiento. Es probable que necesite ver a su médico con frecuencia después de su tratamiento. Pregunte acerca de qué tipo de programación puede esperar. Si usted está tomando algún medicamento, continué tomándolo según lo prescrito por su médico . Tome nota de cualquier efecto secundario e infórmeselo a su médico. Si se ha sometido a cirugía de ovario u a otros tratamientos, siga la instrucciones que se le han proporcionado. También puede ser necesario hacer algunos de los siguientes cambios saludables a su estilo de vida: no fume y limite la cantidad de alcohol que bebe. Ambas actividades pueden aumentar su riesgo de cáncer. Coma una dieta saludable con más frutas, vegetales y granos enteros. Una dieta saludable puede reducir el riesgo de que el cáncer regrese. Manténgase físicamente activo. El ejercicio y las actividades diarias pueden hacer que se sienta mejor. Es importante hablar con su médico antes de comenzar un plan de ejercicios o hacer cambios de estilo de vida. Tenga en cuenta que algunos tratamientos contra el cáncer pueden hacer que se sienta muy cansado. Pregúntele a su médico qué puede hacer para sentirse menos cansado . Saber que tiene cáncer puede ser abrumador. Es posible que tenga preocupaciones sobre su afección y cómo afecta a su familia, sus tratamientos y estancias en el hospital, gastos médicos y su trabajo. Afortunadamente, hay maneras de lidiar con esto. Hable con su equipo de atención médica, ellos pueden ayudarle a encontrar el soporte que sea adecuado para usted. Las siguientes fuentes de apoyo pueden ayudarle a abordar sus preocupaciones: trabajadores, sociales, líderes religiosos, consejeros y grupos de apoyo. Hable con su médico si tiene alguna pregunta sobre su plan de tratamiento, medicamentos o cambios de estilo de vida que la ayuden a manejar el cáncer de ovario.
ANH15162es
7
views
Comprender el cáncer de ovario
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
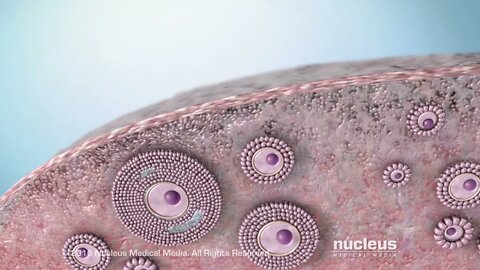
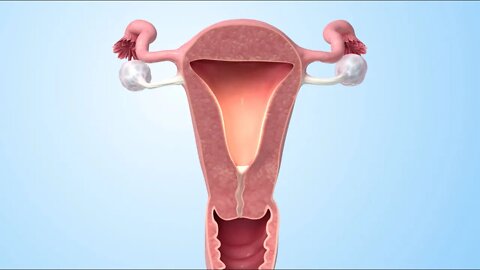
MEDICAL ANIMATION TRANSCRIPT: Usted o alguien que usted quiere ha sido diagnosticada con cáncer de ovario. Este video le ayudará a entender la enfermedad y cómo puede afectarle. Las mujeres tienen dos ovarios. Estos se encuentran a ambos lados del útero. Los ovario son la parte del sistema reproductivo que produce y almacena los óvulos. Los ovarios también producen las hormonas estrógeno y progesterona. Las dos trompas de Falopio están unidas al útero. Después de que un óvulo sale del ovario, pasa a través de una de las trompas hasta el útero. Los ovarios se encuentran cerca del peritoneo. El peritoneo es el revestimiento interno del abdomen que también cubre la mayor parte de los órganos del abdomen y la pelvis. La mayoría de los cánceres de ovario comienzan en la capa externa del ovario. Esto se conoce como cáncer de ovario epitelial. El cáncer ocurre cuando las células crecen fuera de control. Las células anormales continúan dividiéndose y pueden diseminarse a otras partes del cuerpo. Los tumores de células germinales son otro tipo de cáncer de ovario. Se inician en las células que producen óvulos. Los tumores del estroma son un tercer tipo. Comienzan en el tejido de soporte de los ovarios, donde se producen las hormonas. Hay varios factores que pueden aumentar el riesgo de cáncer de ovario epitelial. Un factor de riesgo es la edad: la mitad de los cánceres de ovario ocurren en mujeres de 63 años o más. La obesidad es otro factor de riesgo: las mujeres con un índice de masa corporal de 30 o más tienen un riesgo más alto. El riesgo también es mayor para las mujeres que tienen su primer hijo después de los 35 años o que nunca han tenido un bebé. Recibir terapia hormonal solo con estrógenos después de la menopausia puede aumentar el riesgo. También los antecedentes familiares de cáncer de ovario y otros tipos de cáncer también puede aumentar el riesgo. Es posible que el cáncer de ovario no presente síntomas tempranos. Los síntomas del cáncer de ovario son a menudo vagos. Cuando aparecen los síntomas, estos pueden incluir dolor en la pelvis o el abdomen, gases o hinchazón, sangrado menstrual irregular, sensación de que siempre se necesita orinar, estreñimiento y dolor de espalda. En la mayoría de los casos, estos síntomas son causados por afecciones que no son cáncer de ovario. Si usted tiene cáncer de ovario, el médico tendrá que determinar los subtipos del carcinoma ovárico, la calificación y la etapa o progresión de la enfermedad para ayudar a planificar su tratamiento. Cada calificación se basa en la diferencia del aspecto de las células cancerosas en comparación con las células de ovario normales, observadas bajo un microscopio. Cuánto mayor sea la calificación, más agresivo es el cáncer. Cada etapa del cáncer de ovario se basa en qué tan lejos se han extendido las células cancerosas desde donde empezaron. La primera etapa significa que el cáncer está solo dentro de los ovarios. En la segunda etapa, el cáncer se ha diseminado a otros órganos, pero todavía está dentro de la pelvis . En la etapa tres, el cáncer se ha diseminado más allá de la pelvis, ya sea hacia al peritoneo o los ganglios linfáticos detrás del peritoneo, o hacia ambos. La cuarta etapa significa que el cáncer se ha diseminado a órganos fuera del peritoneo. Si se enfrenta a un diagnóstico de cáncer de ovario, continúe conversando con su médico y su equipo de atención del cáncer.
ANH15160es
7
views
Implantes de progestina
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Alrededor de una vez al mes, ocurre un conjunto de eventos conocido como el ciclo menstrual que prepara el cuerpo de una mujer para el embarazo. Los cambios de los niveles de sustancias químicas naturales en el flujo sanguíneo, las hormonas controlan estos eventos. Los órganos reproductores afectados por estas hormonas incluyen la vagina, el cuello uterino, el útero, las trompas de Falopio y los ovarios. Los ovarios producen dos hormonas principales llamadas estrógeno y progesterona. A medida que el nivel de estrógeno comienza a aumentar hace que la mucosidad del interior del cuello uterino que normalmente es espesa se vuelva más líquida. El estrógeno también activa otras hormonas para provocar que uno de los ovarios libere un óvulo. Este proceso se llama ovulación. Si una mujer tiene relaciones sexuales durante este periodo las células reproductivas del hombre llamadas espermatozoides puede atravesar la mucosa más delgada y fecundar un óvulo. En el útero el estrógeno hace que el endometrio se vuelva más grueso preparándolo para recibir un óvulo fecundado. El aumento de los niveles de progesterona hace que las glándulas del endometrio liberen un líquido que alimenta el ovulo fecundado. La progesterona también hace que la mucosidad del cuello uterino que se había puesto más líquida se vuelva hacer más densa para impedir el paso de los espermatozoides. Si el óvulo no ha sido fecundado los niveles de estrógeno y progesterona comienzan a disminuir, esta disminución de los niveles de hormonas provoca la menstruación, un proceso en el que el útero expulsa por la vagina sangre y el tejido que recubre el interior del útero. Un implante de progestina es un método anticonceptivo, previene el embarazo hasta por tres años. El dispositivo se implanta debajo de la piel en el antebrazo de la mujer. Un implante de progestina es una varilla de plástico flexible que tiene el tamaño de un fósforo y contiene una hormona similar a la progesterona llamada progestina. Está diseñado para ser visible en las radiografías. El implante libera continuamente progestina en el cuerpo. Los niveles altos de progestina ayudan a evitar que el cuerpo libere otras hormonas que causan la ovulación. Sin ovulación no hay óvulo disponible para la fecundación, por lo que la mujer no puede quedar embarazada. Una segunda manera en que la progestina evita el embarazo se relaciona con su efecto sobre la mucosidad del cuello uterino. Los niveles altos de progestina mantiene la mucosa del cuello uterino lo suficientemente gruesa como para que el esperma no pueda atravesarla. Una tercera forma en que la progestina evita el embarazo es a través de su influencia en el recubrimiento uterino. A diferencia de la progesterona natural, la progestina tiene una composición química ligeramente diferente. Con el tiempo hace que el recubrimiento uterino sea más delgado en lugar de más grueso. Como resultado, si se llegara a producir la fecundación de un óvulo, el endometrio puede ser demasiado delgado como para permitir su permanencia en el útero por lo que se produce la expulsión fuera del cuerpo en el siguiente periodo menstrual. Un médico puede insertar un implante de progestina durante una visita al consultorio. Para ello el médico hace dos marcas con un marcador estéril, una para el sitio de inserción y otra que utiliza como guía para la dirección durante la inversión. Posteriormente, el área de inserción se limpia con un antiséptico y se adormece con un anestésico local. A continuación, el médico utiliza un aplicador especial para punzar la piel con una aguja hueca que con contiene el implante y deslizarlo debajo de la piel. Finalmente, retira la aguja dejando el implante dentro. Después de colocar el implante, el médico se asegura de que la ubicación sea correcta palpando ambos extremos del implante debajo de la piel. Se aplica un vendaje sobre el lugar de la inserción. El sangrado menstrual con un implante de progestina puede incluir cambios en la frecuencia del sangrado, la intensidad del sangrando y la duración de cada período de sangrado. Aunque es posible retirarlo antes, el implante se debe quitar después de los tres años. El médico puede reemplazarlo por un implante nuevo en ese momento. Si hay sospecha de embarazo, es importante que la mujer consulte con su médico. Es posible que sea necesario retirar el implante. Un implante de progestina tiene más del 99% de eficacia en la prevención del embarazo, esto significa que menos de una de cada cien mujeres que utilizan este método anticonceptivo quedan embarazadas por año. Para obtener más información sobre cualquier tipo de método anticonceptivo hable con su médico.
ANH16183es
9
views
Biology Quiz: True or false?Animal cells have a cell wall.
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Animal cells never have a cell wall.
35
views
Biology Quiz: What structure gives plant cells shape, support and protection?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Plant cells also have a cell wall outside of their cell membranes that shape, support and protect the plant cell.
18
views
Biology Quiz: What makes chloroplasts green?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
It's green because it has a green pigment, called chlorophyll.
21
views
Biology Quiz: In what organelle does photosynthesis take place?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
The chloroplast is where photosynthesis happens.
15
views
Lupus
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: El lupus es una enfermedad en la cual el sistema inmunológico ataca a los tejidos de su propio cuerpo. El sistema inmunológico esta compuesto de glóbulos blancos que defienden al cuerpo ante invasores extraños. Normalmente cuando invasores extraños como virus y bacterias ingresan al cuerpo, algunos de los glóbulos blancos los identifican como extraños porque contienen sustancias llamadas antígenos. Este reconocimiento provoca que algunos de los glóbulos blancos produzcan marcadores químicos llamados anticuerpos. Estos anticuerpos se adhieren a los antígenos marcando los invasores extraños como objetivos en el sistema inmunológico para destruirlos. En una enfermedad autoimmune como el lupus, los glóbulos blancos hacen anticuerpos erróneamente para marcar algunas de las células de su propio cuerpo y circular productos químicos para la destrucción. Cuando los anticuerpos se adhieren a sus células normales, los glóbulos blancos comienzan a atacarlos como si fueran antígenos extraños. Con el tiempo esta respuesta inmune provoca inflamación en las partes del cuerpo donde sea que ocurra este ataque. La inflamación hace que los vasos sanguineos en el área afectada se expandan y derramen fluido provocando enrojecimiento e hinchazón. Los síntomas del lupus ocurren a medida que el sistema inmunológico comienza a atacar diferentes áreas del cuerpo. Usted puede desarrollar un exantema en alas de mariposa en la piel de las mejillas y la nariz. Pueden formarse pequeñas llagas o ulceras dentro de la nariz y la boca. Usted podría desarrollar artritis en la cual las articulaciones se inflaman y se hinchan. Cuando los dedos están fríos pueden volverse de color azul o blanco, a esto se le conoce como fenómeno de Raynaud. Podría sentir dolor durante una respiración profunda provocada por la pleuresía, una afección en la cual el recubrimiento externo de los pulmones está inflamado. Los glóbulos blancos pueden crear anticuerpos que se adhieren a partes de su ADN de células gastadas. El par adherido llamado complejo inmune puede albergarse en los riñones y provocar inflamación, esta afección se conoce como nefritis lúpica. La recaída de cualquiera de sus síntomas incluso aquellos no relacionados con la piel puede deberse a una reacción inusual a la luz solar, llamada fotosensibilidad. Otros síntomas comunes del lupus incluyen cansancio extremo, dolores de cabeza, fiebre, disminución de los glóbulos rojos en la sangre, conocida como anemia, hinchazón de las piernas, pies, manos, o la piel alrededor de los ojos, dolor muscular y debilidad, y perdida de cabello. Al lupus aveces lo llaman el gran imitador, porque sus diversos síntomas se encuentran en otros poblemas de salud. No hay cura para el lupus, sin embargo usted puede prevenir o reducir las recaídas al evitar la luz directa del sol y utilizar bloqueador solar en el exterior. El bloqueador solar debe tener un factor de protección solar de 55 o mayor y protejer contra la radiación ultravioleta A y la radiación ultravioleta B. Para tratar sus síntomas su médico puede recomendarle una combinación de medicamentos para reducir la inflamación, como los medicamentos antiinflamatorios no esteroideos o AINE y medicamentos contra la malaria. Su médico también podría recomendarle medicamentos que suprimen o previenen el sistema inmunológico ataque a los tejidos de su cuerpo como los corticosteroides.
ANH13101es
83
views
Biology Quiz: What is the term for organisms that capture sunlight for energy?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Some organisms, such as plants, that are photoautotrophic — meaning they capture sunlight for energy — have cells with an organelle called a chloroplast.
13
views
Catalysts and Enzymes
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#catalysts #enzymes #ActivationEnergy
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to talk about catalysts and enzymes. To understand enzymes, we need to know that chemical reactions require energy to occur. Recall that chemical reactions break the chemical bonds in reactants and rearrange those bonds to make products. Let's look at an energy diagram to see how energy changes as a chemical reaction progresses. At the start of a reaction, the reactants will have some amount of energy. If the products end up with more energy than the reactants had at the beginning, then this means the reaction absorbed energy from the environment. This is called an endothermic reaction. However, if the products end up with less energy than the reactants, then this means the reaction released energy. This is called an exothermic reaction. Either way, notice the peak in the energy hill just before the products begin to form. This peak represents the minimum energy the reactants required for the reaction to take place. The reactants need this energy to break their chemical bonds so that different bonds can be formed to make new products. This minimum amount of energy required for a reaction to take place is called activation energy. If the activation energy isn't reached, no reaction takes place. Sometimes, scientists need to make a reaction happen faster. If you want to speed up a reaction, you could increase the energy of the reactants by increasing their temperature, or you could add something called a catalyst. A catalyst is a substance that catalyzes or speeds up a chemical reaction without it being changed or used up by the reaction. Catalysts work by lowering the activation energy needed for the reaction to take place. By requiring less energy, catalysts make reactions happen faster and more efficiently because more of the reacting particles are likely to have sufficient energy or activation energy. And the higher the catalyst concentration, the faster the reaction takes place. Since catalysts aren't changed or used up by a reaction, they're available to catalyze more reactants. Chemical reactions are done in labs, but many chemical reactions also happen constantly inside your body. In living organisms, many reactions require catalysts. Biological catalysts are called enzymes. An enzyme is always made up of proteins. Proteins are one of the four types of organic compounds. We'll discuss them in more detail separately. Enzymes make life possible by lowering the amount of activation energy needed and, therefore, speed up reactions that would otherwise take too long. You need enzymes to stay alive. A reactant acted upon by an enzyme is called a substrate. Each enzyme has a unique active site that only a certain substrate can attach to, like a very specific puzzle piece. When attached, they form what is called an enzyme-substrate complex. This fit between an enzyme and substrate is so specific that it's often compared to a lock and key. When the substrate attaches to the enzyme, the enzyme lowers the activation energy needed to break the bonds in the substrate. Once the bonds break in the substrate, it separates into multiple components called products, which leave the active site. Afterward, the enzyme is still available to participate in another reaction with the same type of substrate. So, what factors affect how well an enzyme works? Well, enzymes work best around particular pH levels and certain temperatures, and the presence or absence of other chemicals can also impact their effectiveness. Not surprisingly, humans and other organisms have adapted to use enzymes that are most effective at the organism's natural temperature and pH values. To sum up, chemical reactions require energy to break the reactants' chemical bonds. The minimum amount of energy required for a reaction to happen is called the activation energy. Catalysts speed up reactions by lowering the activation energy. Catalysts aren't used up by chemical reactions. Biological catalysts are called enzymes. Enzymes are made of proteins. Enzymes act on reactants called substrates. Substrates bind to unique locations called active sites on enzymes. And factors that affect how well an enzyme works include pH, temperature, and other chemicals.
NSV16023
41
views
Can You Answer ALL 28 Questions on Cell Biology? (Everyone misses #22)
If you are an educator at K-12, college, university or medical school, you may download 8 FREE medical animations from Nucleus by signing up for a free trial: http://nmal.nucleusmedicalmedia.com/free-trial-membership-a
23
views
Biology Quiz: What are the 2 components of the cytoskeleton?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
The cytoskeleton includes the threadlike microfilaments, which are made of protein, and microtubules, which are thin, hollow tubes.
14
views
Biology Quiz: What is the main function of the cytoskeleton?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Meanwhile, the cell maintains its shape through a cytoskeleton.
14
views
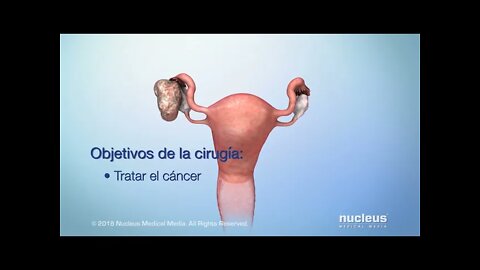
Opciones de tratamiento para el cáncer de ovario
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Usted o alguien que usted quiere puede haber sido diagnosticada con cáncer de ovario. Este video le ayudará a entender las opciones de tratamiento disponibles. El cáncer de ovario comienza en los ovarios, que son la parte del sistema reproductivo que produce y almacena los óvulos. El cáncer ocurre cuando hay células anormales que crecen sin control. Las células cancerosas continúan dividiéndose y pueden expandirse. Su tratamiento se basa en el tipo y etapa del cáncer, y en otros factores importantes. Los principales tratamientos para el cáncer de ovario son la cirugía y la quimioterapia. Su plan de tratamiento puede incluir ambas opciones. También puede recibir uno o más de los siguientes tratamientos: terapia dirigida, radioterapia o terapia hormonal. La cirugía para extirpar el tumor es a menudo el primer paso en su plan de tratamiento. El objetivo de la cirugía es tratar el cáncer mediante la extirpación de la mayor cantidad del tumor que sea posible. Si usted tiene cáncer de ovario epitelial, el objetivo es también determinar la etapa del cáncer. La cirugía incluye la extirpación de uno o ambos ovarios. Las trompas de Falopio afectadas también pueden ser extirpadas. Si se extirpa el útero, la cirugía puede incluir la extirpación del cuello uterino. Para eliminar la mayor cantidad posible de cáncer, el cirujano puede necesitar tomar tejidos de órganos que no están en el sistema reproductivo. Pueden tomarse ganglios linfáticos o el omento, que es el delantal de tejido que cubre el estómago y el intestino. La cirugía también puede implicar tomar tejido de órganos cercanos como la vejiga, el estómago o el colon. La quimioterapia utiliza medicamentos para combatir el cáncer. Los medicamentos entran en el torrente sanguíneo y viajan por todo el cuerpo. La quimioterapia funciona destruyendo las células cancerosas o impidiendo su multiplicación. Las células sanas del cuerpo también pueden verse afectadas, lo que puede producir efectos secundarios. La terapia dirigida es un tratamiento para el cáncer epitelial de ovario avanzado. Se dirige a la parte de las células cancerosas que las hace diferentes de las células normales. Estos medicamentos cambian la forma en que las células cancerosas se dividen, crecen y se reparan. Como resultado, el tumor puede dejar de crecer, o incluso reducir el tamaño. Las células cercanas y sanas del cuerpo también pueden verse afectadas, lo que lleva a los efectos secundarios. Otros tratamientos para el cáncer de ovario son la radioterapia y terapia hormonal. Estos se utilizan con menos frecuencia. Su médico puede recetar una combinación de estos tratamientos. Discuta con su médico cualquier pregunta que tenga sobre su plan de tratamiento o efectos secundarios. Asegúrese de tomar sus medicamentos según las indicaciones de su médico.
ANH15161es
8
views
Nucleic Acids
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#NucleicAcids #DNA #RNA
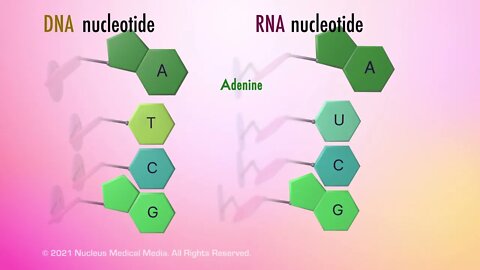
SCIENCE ANIMATION TRANSCRIPT: The final organic macromolecule we'll cover is nucleic acids. A nucleic acid is an organic macromolecule, which means it's a large organic compound made from thousands or hundreds of thousands of smaller molecules. There are two types of nucleic acids, deoxyribonucleic acid or DNA and ribonucleic acid or RNA. Notice that both terms actually contain the words nucleic acid in them. In living organisms, DNA is the main component of chromatin, which will condense into the familiar chromosome shape prior to cell division. The DNA in chromosomes is organized in a specific order that makes up an organism's genes. Genes contain the directions for every function, trait, and activity in a living organism. These activities include growth, reproduction, and especially, heredity, which means the ability to pass on genes to offspring. The other type of nucleic acid, RNA, is integrally involved in building specific proteins by assembling their amino acids in the correct order. Proteins are responsible for all cellular functions in living organisms. So what are nucleic acids made of? Of course, as an organic macromolecule, nucleic acids contain the element carbon. Like proteins, nucleic acids also contain hydrogen, oxygen, and nitrogen. But nucleic acids also contain the element, phosphorous. So what is the structure of nucleic acids? Well, both DNA and RNA are made up of monomers called nucleotides. All nucleotides contain three compounds. The first compound is a phosphate group which contains phosphorous. The second compound is a five-carbon sugar. All five-carbon sugars can be called a pentose. A good way to remember this is that pent means five and the suffix, -ose, means sugar. The specific pentose sugar in RNA is called ribose. The pentose sugar in DNA has one less oxygen than ribose in RNA, which leads to its name, deoxyribose. The third compound in a nucleic acid nucleotide is a weak base that always contains nitrogen. For this reason, this compound is referred to as a nitrogenous base. The nucleotides in DNA contain one of four possible nitrogenous bases. They are called adenine, thymine, cytosine, or guanine. These bases are usually abbreviated as A, T, C, and G. Similarly, the nucleotides in RNA also contain one of four nitrogenous bases, adenine, uracil, cytosine or guanine. They are abbreviated as A, U, C, and G. Notice that both DNA and RNA have adenine, cytosine, and guanine but only DNA has thymine and only RNA has your uracil. So how do nucleotide monomers assemble into nucleic acids? Well, the phosphate group in one nucleotide bonds with the pentose sugar in another nucleotide. The assembled string of nucleotide forms a nucleic acid polymer. Even though both RNA and DNA have alternating sugar and phosphate groups, there are differences in their overall structure. For example, RNA is a single-stranded nucleic acid while DNA is a double-stranded nucleic acid. The two strands in DNA are linked by a hydrogen bond that connects the nitrogenous bases from one strand to the nitrogenous bases from the other strand. In summary, nucleic acids are organic macromolecules. The two types of nucleic acids are DNA and RNA. DNA and RNA are used to transmit hereditary information and to instruct the cell how to properly construct proteins. Nucleic acids contain carbon, hydrogen, oxygen, nitrogen, and phosphorous. Nucleotides are the monomers that bond together to form DNA or RNA. Nucleotides consist of three compounds, a phosphate group, a five-carbon sugar, also known as a pentose, and a nitrogenous base. The nitrogenous bases in DNA are adenine, thymine, cytosine, and guanine. The nitrogenous bases in RNA are adenine, uracil, cytosine, and guanine. The five-carbon sugar in DNA is called deoxyribose while the five-carbon sugar in RNA is called ribose.
NSV16033
77
views
Proteins
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#proteins #AminoacidMolecule #peptides
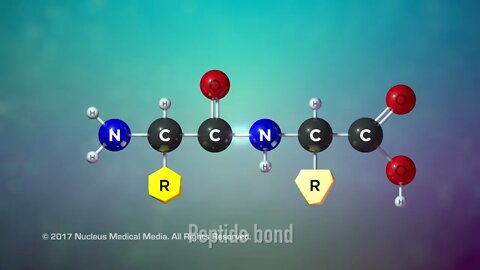
SCIENCE ANIMATION TRANSCRIPT: So far we've covered two of the organic macromolecules: carbohydrates and lipids. Today, we'll be talking about proteins. Proteins have many uses. For example, many different proteins are used to form the majority of various body tissues including muscle, bone, cartilage, and hair. Other proteins form specialized channels in cell membranes to let certain substances into or out of the cell. Immunoglobulins, also known as antibodies, are made of proteins. They are an important part of the immune system. Antibody proteins label foreign substances in the body so that the immune system can identify and destroy them. And all living organisms use regulatory proteins called enzymes to speed up chemical reactions in the body. So what are proteins made of? Well, all proteins are organic macromolecules, which means they're large molecules containing the element carbon. Like carbohydrates and lipids, proteins also contain hydrogen and oxygen. In addition, proteins also contain the element nitrogen. So what is the structure of proteins? Well, the protein building block, or monomer, is something called an amino acid molecule. Every amino acid has a particular structure. They all have the same amino group on one side and a weak acid called a carboxyl group on the other. But each amino acid also has a unique side chain called an R-group. It's made up of one or more atoms that distinguish one amino acid from another. There are only 20 possible different R-groups, so that means there are only 20 possible different amino acids. A protein is created when amino acids chemically bond to each other. The bonds that connect amino acids to make a protein are called peptide bonds. Small protein molecules are sometimes referred to as protein polymers or peptides. Like interconnecting toy blocks, amino acids can join together to form even longer protein polymers called polypeptides. Changing the order of the amino acid sequence or the length of the polypeptide is what creates countless different proteins. In addition, a protein's final folded shape is critical to the function it performs. Heat or chemicals can change a protein's shape but not its amino acid order. However, the change in a protein's shape can cause the protein to lose its functionality. This is called denaturing a protein. Denaturing is usually not reversible. An example of denaturing a protein happens when you fry an egg. Egg whites are pure protein. If you heat an egg, the protein in the egg white becomes denatured. You can see the result of this denaturing as the egg white changes from a clear-colored liquid to a white-colored solid. Even after cooling, the egg whites' denatured proteins can't change back to their original shape. So the egg white retains its opaque white color and solid form. To sum up, proteins are organic macromolecules containing carbon, hydrogen, oxygen, and nitrogen. Enzymes, most body tissues, and many cells in the immune system are all made up of proteins. Amino acids are the monomers that make up proteins. There are only 20 different amino acids. Small protein polymer molecules are often called peptides. Long chains of amino acids are also a type of protein polymer called polypeptides. Proteins differ in amino acid sequence, polypeptide length, and folded shape. A protein's folded shape is critical to its function. ♪ [music] ♪
NSV16038
38
views
Biology Quiz: Cells needing more energy require more of which organelles?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
Cells that need more energy have more mitochondria.
3
views
Lipids
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#lipids #fats #steroids
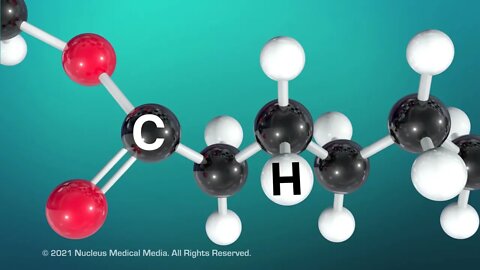
SCIENCE ANIMATION TRANSCRIPT: Today, we're going to talk about lipids. Lipids are an integral part of every cell membrane in every living organism. Looking closer, you can see that lipids are part of the phospholipid membrane that is the boundary of every single cell. Most people think of lipids as fats, such as the fat in your body. Fats provide long-term energy storage and insulation in living organisms. But fats are just one type of lipid. Oils, waxes, and steroids are also types of lipids. Examples of steroids include cholesterol and hormones such as testosterone, which is produced in the testicles, and estrogen, which is produced in the ovaries. A common feature of all lipids is that they don't dissolve in water. So what makes something a lipid? All lipids are organic macromolecules. This means lipids are large molecules containing the element carbon. Lipids also contain hydrogen and oxygen. Organic macromolecules, such as lipids are formed by many units called monomers that are chemically bonded together. In lipids, the typical monomer is something called a fatty acid. A fatty acid contains a chain of carbon atoms attached to each other. Hydrogen atoms are also attached to these carbon atoms. You may recall that carbon can form up to four covalent bonds with other atoms. When each carbon atom forms two single bonds with adjacent carbon atoms and another two single bonds with adjacent hydrogen atoms, we call this fatty acid saturated. This means the fatty acid is saturated with all the hydrogen atoms it can possibly contain. Because of this structure, saturated fatty acids are straight molecules that can pack tightly together. As a result, saturated fats are usually solid at room temperature. Examples of saturated fats include lard and butter. In contrast, unsaturated fats are usually liquid at room temperature. These include things like vegetable oil and olive oil. So why are unsaturated fats liquid at room temperature? Well, unsaturated fatty acids have one or more double covalent bonds between carbon atoms. This means the unsaturated fatty acid has gaps in the hydrogen saturation. The carbon double bonds form kinks in the fatty acid, which prevent the molecules from packing together tightly. As a result, the more loosely packed molecules form a liquid instead of a solid at room temperature. Here's a tip to help you remember which type of fat is solid or liquid. Use the letter S at the beginning of the word saturated for solid at room temperature. Now that we've talked about fatty acids as lipid monomers, what is a lipid polymer? A lipid polymer, called a triglyceride, is formed when three fatty acids bond to a glycerol molecule. A triglyceride is saturated if it contains only saturated fatty acids. And a triglyceride is unsaturated if it contains any unsaturated fatty acids. To sum up, all living organisms use lipids in all of their cell membranes, as well as for long-term energy storage and insulation and in hormone formation. Lipids are organic macromolecules containing mostly carbon atoms, as well as hydrogen and oxygen atoms. Lipid monomers are fatty acids. Saturated fatty acids are saturated with hydrogen because their carbon atoms form only single bonds. As a result, saturated fats are solid at room temperature. In contrast, unsaturated fatty acids have gaps in their hydrogen saturation, because their carbon atoms form one or more double bonds. As a result, unsaturated fats are liquid at room temperature. Lipid polymers are called triglycerides. Saturated triglycerides contain only saturated fatty acids. Triglycerides are called unsaturated if they contain any unsaturated fatty acids.
NSV16036
64
views
Biology Quiz: What energy-carrying molecule is produced during cellular respiration?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
4
views
Carbohydrates
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#carbohydrates #saccharides #polysaccharides
SCIENCE ANIMATION TRANSCRIPT: In this video, we'll discuss carbohydrates. A carbohydrate is an organic macromolecule, which means it's a large molecule containing the element carbon. The hydrate part of carbohydrates means it also contains the components of water, hydrogen and oxygen. Carbohydrates always contain these elements in a proportion of one carbon atom, to two hydrogen atoms, to one oxygen atom. Think of it this way. All carbohydrate molecules will always have an equal number of carbon and oxygen atoms and will also always have twice as many hydrogen atoms. So why are carbohydrates important? Carbohydrates are the main fuel source from which all living things get their energy, but carbohydrates don't always have the same overall structure. They are classified as either simple or complex based on their chemical structure. All simple carbohydrates are sugars, referred to as saccharides. A saccharide can be made of one sugar molecule, called a monosaccharide, or two sugar molecules, called a disaccharide. An example of a monosaccharide is the sugar glucose. It's a monosaccharide because it's made of a single sugar molecule. Simple sugars like glucose are the quickest form of energy because your cells can break them down easily. Words ending in O-S-E or OSE are typically sugars, such as the monosaccharides fructose, galactose, and dextrose. Like glucose, these monosaccharides are all simple sugars because they are made up of a single sugar molecule. When glucose and fructose are chemically bonded together, we get a disaccharide called sucrose, commonly known as table sugar. Sucrose is a disaccharide because it's made up of two monosaccharides. Like monosaccharides, disaccharides such as sucrose also end in O-S-E because they're sugar molecules. Now let's talk about complex carbohydrates, also known as polysaccharides. Polysaccharides are very long chains of multiple monosaccharides chemically bonded together. An entire polysaccharide is referred to as a polymer, while each individual monosaccharide in this long chain is referred to as a monomer. Examples of polysaccharides include cellulose, starches, and glycogen. Cellulose is found in all plants. It's a structural carbohydrate that is found in the cell wall of every plant cell. Starches are found in many of the foods we eat, such as potatoes and corn. Starches are also found in grains such as wheat and rice. The body's digestive system breaks down starches into glucose molecules to fuel the activities of all of our cells. Extra glucose molecules that the body doesn't immediately need for energy are stored as polysaccharides called glycogen either in the liver or in skeletal muscles. To sum up, carbohydrates are organic macromolecules containing carbon, hydrogen, and oxygen in a one to two to one proportion. Carbohydrates are the main source of energy in living organisms. Carbohydrate monomers are simple sugars called monosaccharides. Disaccharides are two monosaccharides bonded together. Monosaccharide and disaccharide names typically end in OSE such as glucose, fructose, and sucrose. Carbohydrate polymers are called polysaccharides. Polysaccharides include cellulose, starches, and glycogen. [music]
NSV16030
26
views
Biology Quiz: What is the term for making ATP to provide energy for the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
5
views
Biology Quiz: What organelle is the “powerhouse” of the cell?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
5
views
Información breve sobre la incontinencia urinaria de esfuerzo (IUE)
Si deseas ver más imágenes médicas en 3D con precisión científica, suscríbase a nuestro canal: https://www.youtube.com/user/nucleushealthvideose
MEDICAL ANIMATION TRANSCRIPT: Esta animacin mdica 3D ofrece una breve descripcin de la incontinencia urinaria de esfuerzo o de la IUE, una visin general de la anatoma y fisiologa del sistema urinario femenino y tres tratamientos quirrgicos para la IUE como una suspensin de la vejiga abdominal abierta (Burch), suspensin de la vejiga laparoscpica y un procedimiento de cabestrillo . El sistema urinario incluye los riones, los urteres, la vejiga y la uretra. En los procedimientos de suspensin de la vejiga, las suturas se colocan en el tejido vaginal cerca del cuello de la vejiga, entonces se une a los ligamentos inguinales. Al cambiar la ubicacin y el ngulo de la uretra, la orina tiene menos de una tendencia a fluir fcilmente a travs de bajo estrs. En un procedimiento de cabestrillo, una correa o arns de material sinttico se adjunta debajo de la uretra y el cuello de la vejiga y las inmediaciones, alrededor de la fascia del msculo recto para formar un soporte hamaca-como por la uretra.
ANH00021es
5
views
Overview of Organic Compounds
For Employees of hospitals, schools, universities and libraries: download up to 8 FREE medical animations from Nucleus by signing up for a free trial at: http://nmal.nucleusmedicalmedia.com/biology_youtube
#OrganicCompounds #OrganicMonemers #polymers
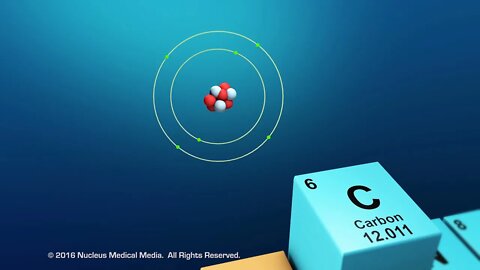
SCIENCE ANIMATION TRANSCRIPT: Today, we are going to be talking about organic compounds. Organic compounds are studied in biology because they are found in all living things. All organic compounds have the element carbon in them. In other words, all living organisms contain carbon. What's so special about the element, carbon? For one thing, no other element matches carbon's unique versatility to bond with other elements. Let's look at why this is true. For starters, carbon has an atomic number of six. That means that every atom of carbon has six protons in the nucleus. As an electrically neutral atom, carbon also has six electrons. Two core electrons are in the first energy level, which means it has four remaining valence electrons in the second energy level. Remember, valence electrons are those electrons available for bonding with other atoms. Accompanying these four electrons are four bonding sites, or four places that carbon can form bonds with other carbon atoms, or with atoms of other elements. Carbon's four valence electrons and four bonding sites allow it to form strong covalent bonds with many other elements, including hydrogen, oxygen, nitrogen, and phosphorus. Another feature of carbon atoms is they often form covalent bonds with other carbon atoms, to a nearly unlimited degree. This means that two carbon atoms can bond to one another, 50 carbon atoms can bond to one another, or even hundreds of carbon atoms can bond to one another. The ability of carbon atoms to bond to one another gives it the unique ability to shorten or lengthen a chain of carbon atoms to meet the very demands of the chemistry of life. So what kinds of molecules can carbon form? Well, small organic molecules called monomers are chemically bonded atoms that always include carbon. In addition to carbon, organic monomers usually contain hydrogen and oxygen, possibly along with nitrogen or phosphorus. Organic monomers often chemically bond to each other, joining together like beads on a string. This string of attached monomers will often continue to chemically bond with additional monomers, creating a much larger molecule, called a polymer. This process is called polymerization. Polymers may be made of different monomers or repeating units of the same monomer. Many organic polymers in the cells of living organisms are such large molecules that they're often referred to as macromolecules. DNA is an example of a macromolecule. Macro molecules can contain hundreds or even thousands of atoms. The four types of organic macromolecules are carbohydrates, lipids, proteins, and nucleic acids. Although they are all very large molecules, each type of organic macromolecule is distinct and different from the others. We'll discuss these four types of macromolecules in more detail separately. To sum up, organic compounds are found in all living things. All organic compounds contain the element carbon. Carbon atoms have a unique ability to bond to other carbon atoms, as well as other elements, such as hydrogen, oxygen, nitrogen, and phosphorus. Organic monomers are chemically bonded atoms that always include carbon. Polymerization is the process of creating long molecules, called polymers, from multiple bonded monomers. And macromolecules are very large organic molecules. [music]
NSV16027
46
views
Biology Quiz: Which organelles take in and break down cellular debris?
Tell us what you think about our new Biology Quiz shorts, and subscribe to our new Biology channel to get more quizzes and biology animations!
#shorts #biology #cells
8
views