Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
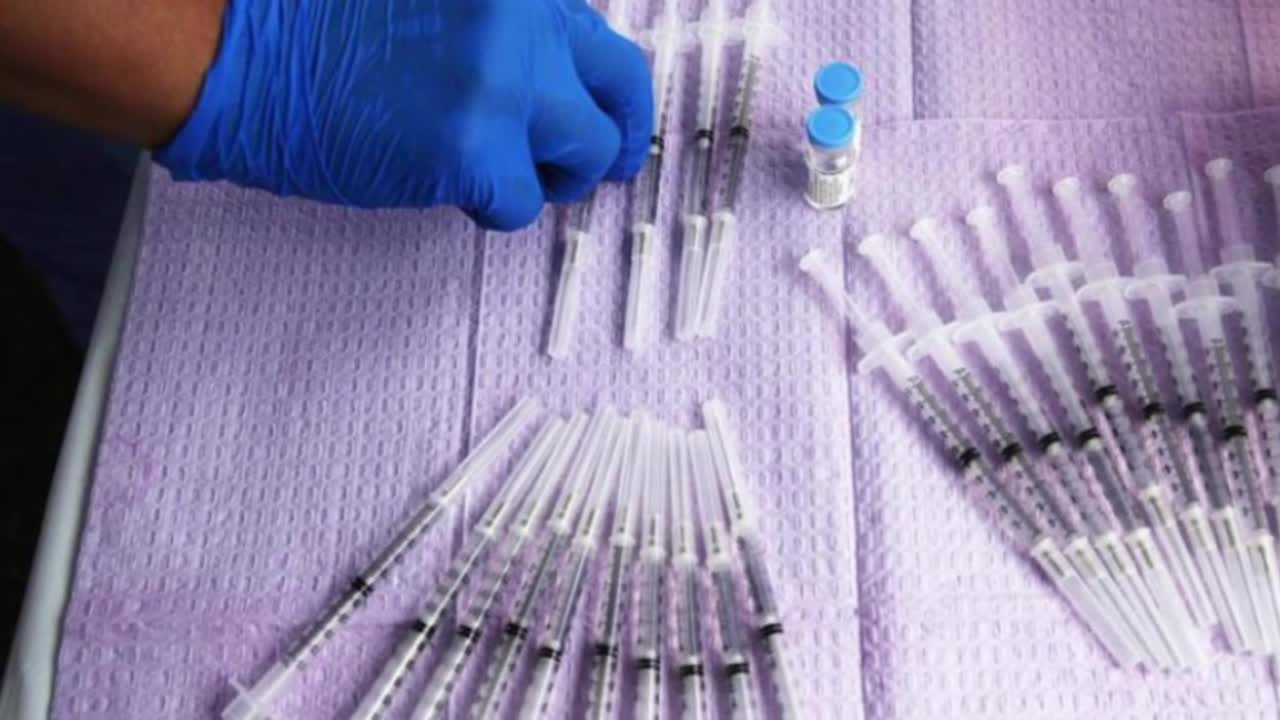
Johnson and Johnson COVID Vaccine on ‘Pause’ After FDA, CDC Report Rare Blood Clot Issues.
Loading 3 comments...
-
 0:51
0:51
New York Post
4 years agoNYC man tests positive for COVID two weeks after Johnson & Johnson vaccine
10.6K194 -
 2:02
2:02
KMTV
4 years agoQuestions and answers about Johnson & Johnson COVID vaccine
8135 -
 3:33
3:33
WSYM
4 years agoJohnson & Johnson releases data on new COVID vaccine
4722 -
 0:30
0:30
KTNV
4 years agoJohnson & Johnson vaccine: FDA report finds single-shot vaccine to be safe, effective
90 -
 2:50
2:50
WXYZ
4 years agoFDA approves Johnson & Johnson vaccine
3791 -
 3:17
3:17
KGTV
4 years agoIn-Depth: J&J vaccine paused after rare blood clots
2332 -
 2:21
2:21
WXYZ
4 years agoIs vaccine hesitancy growing after new Johnson & Johnson developments?
691 -
 0:45
0:45
New York Post
4 years agoBiden administration aware of Johnson & Johnson vaccine issues two weeks ago
6152 -
 0:29
0:29
Newsy
4 years agoFDA Will Review Johnson & Johnson Vaccine
1.24K5 -
 1:46
1:46
WMAR
4 years agoJohnson & Johnson vaccine approved
1.23K1