Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.

Approval versus authorization: All three vaccines still being evaluated by FDA
Loading 2 comments...
-
 0:19
0:19
KMGH
2 years agoAdventHealth Littleton adding heart & vascular tower
876 -
 0:55
0:55
WFTX
5 years agoThree vaccines await FDA approval
7 -
 0:15
0:15
WFTX
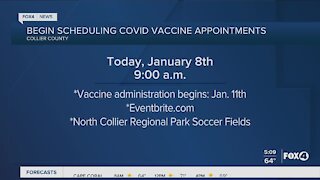
4 years agoCollier County vaccines being scheduled
35 -
 0:46
0:46
KTNV
4 years agoSome COVID vaccines expiring before being administered
1431 -
 2:57
2:57
WFTX
4 years agoThe difference between the three newest covid vaccines
1.06K6 -
 1:16
1:16
KSHB
5 years agoMail-in ballots still being processed
1.62K -
 0:51
0:51
KMGH
4 years agoJeffco Public Health says senior vaccines being handled by Stride
57 -
 1:50
1:50
KTNV
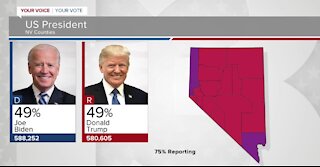
5 years agoVotes still being counted in Nevada
4521 -
 59:02
59:02
The Soul of the EveryMan
5 years agoSoul of the Everyman - Being versus Doing
37 -
 1:58
1:58
KGUN
5 years agoPima County ballots still being counted
507