Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
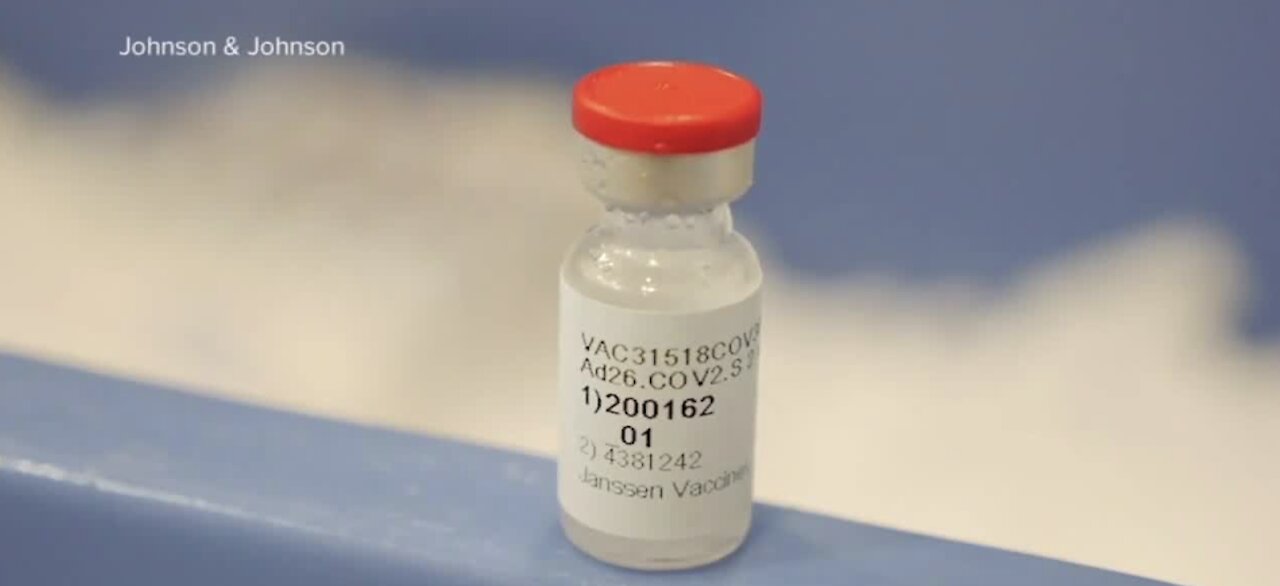
Johnson & Johnson submits its one-shot COVID-19 vaccine for FDA authorization
Loading comments...
-
 2:04
2:04
KTNV
1 year agoMetro police ramp up recruitment efforts amid staffing shortages
1.88K -
 3:24
3:24
KGTV
4 years agoIn-depth: New data on Johnson & Johnson COVID-19 vaccine
1.29K1 -
 1:51
1:51
WFTS
4 years agoPfizer submits Emergency Use Authorization request to FDA for its COVID-19 vaccine
163 -
 1:59
1:59
KSHB
4 years agoJohnson & Johnson single-shot COVID-19 vaccine could boost vaccination rate
757 -
 1:34
1:34
Reuters
4 years agoJ&J files for U.S. COVID-19 vaccine authorization
415 -
 2:35
2:35
KTNV
4 years agoSecond COVID-19 vaccine nears authorization
391 -
 0:41
0:41
Newsy
4 years agoJohnson & Johnson Says One-Shot COVID Vaccine Candidate Is Effective
3.06K7 -
 2:19
2:19
Newsy
4 years agoJohnson & Johnson Covid-19 Vaccine On Hold
1.48K6 -
 0:43
0:43
Newsy
4 years agoJohnson & Johnson Begins Phase 3 COVID-19 Vaccine Trials
1.28K2 -
 2:14
2:14
WFTS
4 years agoUS approves authorization of Pfizer's COVID-19 vaccine
23