Pfizer submits Emergency Use Authorization request to FDA for its COVID-19 vaccine
3 years ago
163
Pfizer announced Friday that it would submit a request to the FDA Friday for its COVID-19 vaccine to be granted Emergency Use Authorization.
Loading comments...
-
 1:12
1:12
WFTS
7 months agoTracking the Tropics | September 5, Evening Update
240 -
 0:33
0:33
WFTX
3 years agoPfizer seeking emergency vaccine use
1.05K3 -
 2:00
2:00
KGUN
3 years agoPfizer seeking emergency use of vaccine
528 -
 2:00
2:00
WCPO
3 years agoPfizer: COVID-19 vaccine 90% effective
2.73K15 -
 2:03
2:03
KGTV
3 years agoPfizer announces COVID-19 vaccine progress
3.52K -
 2:43
2:43
KTNV
3 years agoPfizer announces more information about COVID-19 vaccine
21.2K4 -
 0:55
0:55
WFTS
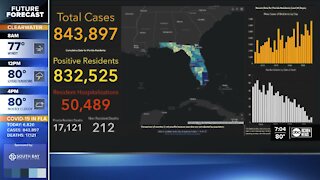
3 years agoPfizer says its COVID-19 vaccine candidate is 90% effective
2.44K2 -
 2:08
2:08
KMGH
3 years agoPfizer says COVID-19 vaccine is looking 90% effective
6501 -
 0:20
0:20
KTNV
3 years agoPfizer to begin testing COVID-19 vaccine in 4 states
3.08K11 -
 1:47
1:47
KSHB
3 years agoKC doctors speak out about Pfizer COVID-19 vaccine trail
7.12K21