Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
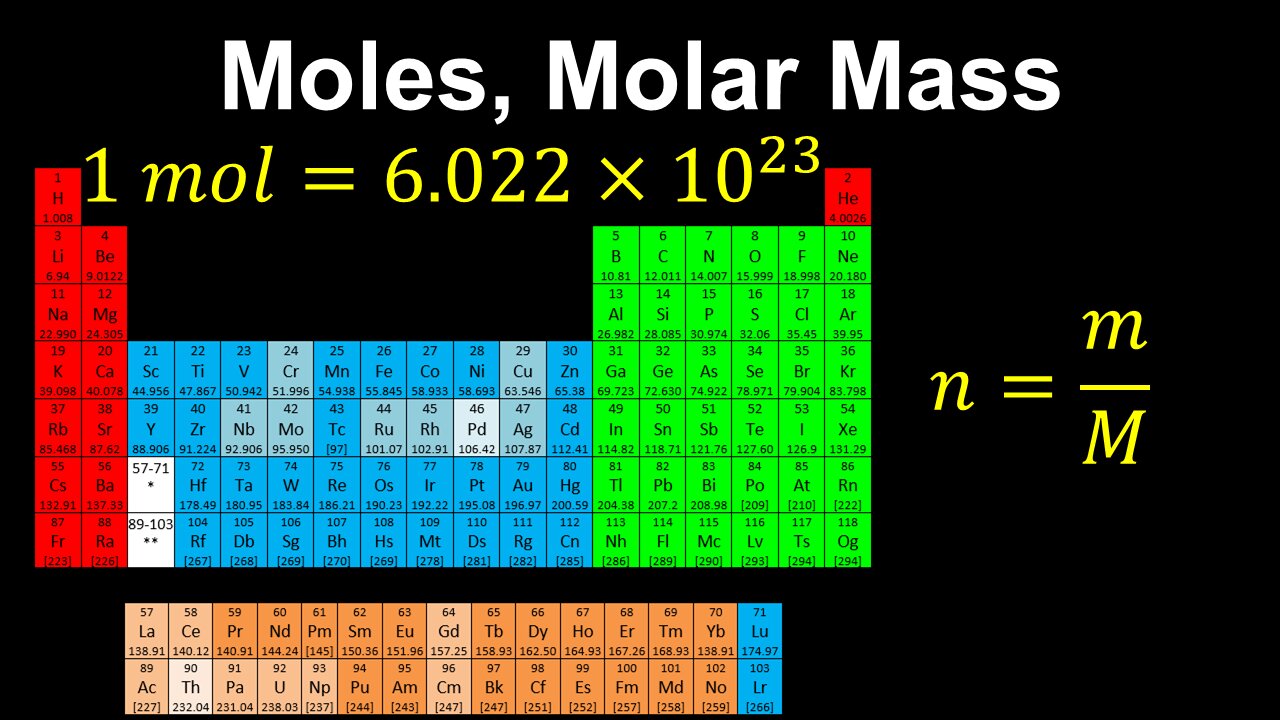
Moles, Molar Mass, Avogadro's Number - AP Chemistry
1 year ago
12
This AP Chemistry video defines the unit of mole using Avogadro's number, which is used to connect the mass of a substance to the number of its particles.
This video is part of Unit 1 of AP Chemistry on Atomic Structure and Properties.
You can contact me through the comments and community section, or email me at drofeng@gmail.com for questions.
Loading comments...
-
 LIVE
LIVE
SpartakusLIVE
7 hours agoSHAVING my BEARD on stream TONIGHT || Chiseled JAWLINE, peak MASCULINITY, and SUAVE FRESH FACE
2,132 watching -
 2:43:56
2:43:56
Barry Cunningham
8 hours agoBREAKING: PRESIDENT TRUMP SPEAKS AT AMERICA'S 250TH ANNIVERSARY KICK-OFF RALLY!
57.6K41 -
 LIVE
LIVE
Repentency
3 hours agoHalo 2 HCS Pro Playing Matchmaking
241 watching -

Alex Zedra
4 hours agoLIVE! CoD tonight :D
11.3K2 -
 1:31:19
1:31:19
AimControllers
2 days ago $6.39 earned🔴LIVE - THE PRE GAME LOBBY PODCAST
47K10 -
 2:17:32
2:17:32
megimu32
5 hours agoOTS: Born to Grill, Raised on Fireworks - 4th of July Throwbacks
26.2K13 -
 LIVE
LIVE
IcyFPS
2 hours ago32 days left in Ranked.. How far do we get?!
326 watching -
 LIVE
LIVE
JdaDelete
1 day ago $0.09 earnedThe Legend Of Zelda Breath of the Wild | July Gone Wild #3
25 watching -
 5:27:59
5:27:59
OhHiMark1776
7 hours ago🟢 07-03-25 ||||| Dur ||||| Baldur's Gate 3 (2023)
6.63K2 -
 13:47
13:47
Adam Does Movies
6 hours ago $0.57 earnedJurassic World Rebirth - Movie Review
12.9K6