Reaction Mechanism and Rate Law, Kinetics - Chemistry
Enjoyed this video? Join my Locals community for exclusive content at
drofeng.locals.com!
1 year ago
6
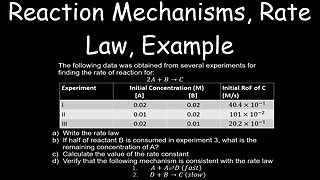
This Chemistry video describes how the rate law can be determined from the slowest (rate determining) step of a reaction mechanism. In this special case, the elementary reactions are irreversible and the first step is the slowest step.
Post your comments/questions below and please subscribe.
#DrOfEng #chemistry #chemicalkinetics
Loading comments...
-
 7:03
7:03
Chemistry - DrOfEng
1 year agoReaction Mechanisms, Rate Law, Example, Kinetics - Chemistry
11 -
 2:12
2:12
Chemistry - DrOfEng
1 year agoReaction Rates, Kinetics - Chemistry
7 -
 8:45
8:45
Chemistry Tutor
4 years agoReaction Kinetics What Makes a Reaction Happen Chemistry for Nurses Lecture Video (Lecture 21)
5 -
 17:20
17:20
TheOrganicChemistryTutor
7 months agoArrhenius Equation Activation Energy and Rate Constant K Explained
100 -
 5:46
5:46
Organic Chemistry I & II
3 years ago $0.03 earnedCarbonyl kinetics
150 -
 1:30
1:30
ScamarcaProductions
7 years agoAnimated short explains Kinetic Molecular Theory
313 -
 5:42
5:42
The Action Lab Archive Channel
1 year agoHow Molecules Can React Slow, But Actually Really Fast-Chemical Reaction Speeds Explained
14 -
 24:00
24:00
Zerofatzreturns on Rumble
11 months agoHow to Read Fast: REACTION
-
 12:07
12:07
Organic Chemistry I & II
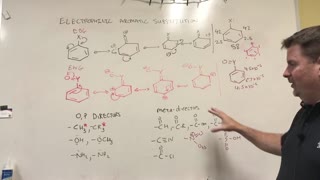
2 years ago $0.12 earnedEAS Rates of Reaction
90 -
 7:26
7:26
Organic Chemistry I & II
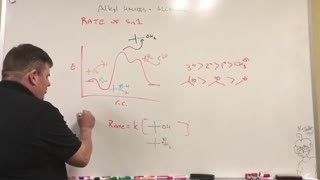
3 years ago $0.17 earnedNucleophilic 1st order (Sn1) Kinetics
195