Premium Only Content
Thermodynamics, example - Physics
Physics video, worked example, thermodynamics problem.
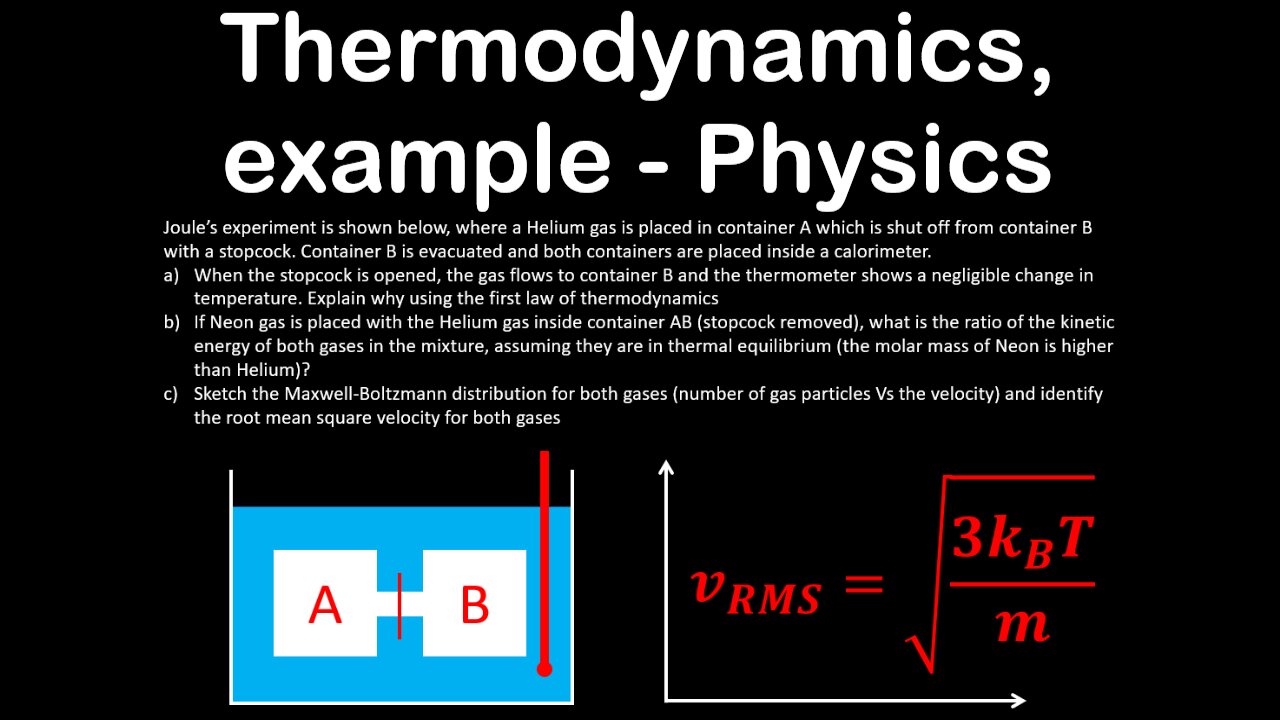
Joule’s experiment is shown below, where a Helium gas is placed in container A which is shut off from container B with a stopcock. Container B is evacuated and both containers are placed inside a calorimeter.
a) When the stopcock is opened, the gas flows to container B and the thermometer shows a negligible change in temperature. Explain why using the first law of thermodynamics
b) If Neon gas is placed with the Helium gas inside container AB (stopcock removed), what is the ratio of the kinetic energy of both gases in the mixture, assuming they are in thermal equilibrium (the molar mass of Neon is higher than Helium)?
c) Sketch the Maxwell-Boltzmann distribution for both gases (number of gas particles Vs the velocity) and identify the root mean square velocity for both gases
Post your comments/questions below and please subscribe.
#physics #thermodynamics #DrOfEng
-
 5:40
5:40
Sugar Spun Run
22 hours ago $0.04 earnedNutella Brownies
171 -
 8:46
8:46
Faith Frontline
14 hours agoBill Maher STUNNED as Charlie Kirk Proves God Exists
812 -
 LIVE
LIVE
FyrBorne
8 hours ago🔴Warzone M&K Sniping: On the Hunt For The Next Fun Builds
8 watching -
 7:11
7:11
MudandMunitions
11 hours agoNY Legal, Still LETHAL! Colt M4 + Griffin Armament GPS3X Prism Sight! NIGHT SHOOT
273 -
 2:11
2:11
WildCreatures
2 days ago $0.32 earnedBrilliant Blue Hyacinth Macaw Eats Nuts With Impressive Dexterity
1.53K4 -
 29:45
29:45
DeVory Darkins
14 hours ago $4.35 earnedDemocrat Governor suffers EMBARRASSING LOSS to Trump as ICE takes Garcia into custody
5.01K51 -
 24:50
24:50
Bitcoin.com
14 hours agoEthereum hit an ATH this weekend | The Weekly Recap for Aug 25
265 -
 56:27
56:27
Actual Justice Warrior
14 hours agoAnti-White Celebrity DESTROYED On Jubilee
1.82K7 -
 19:44
19:44
itsSeanDaniel
1 day agoLiberal Karen INSTANTLY REGRETS Interrupting Putin's Right Hand Man
2088 -
 1:20:38
1:20:38
BlaireWhite
2 days agoThe Dark Truth About UFOs: Why The Government Is Lying
9.78K10