Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
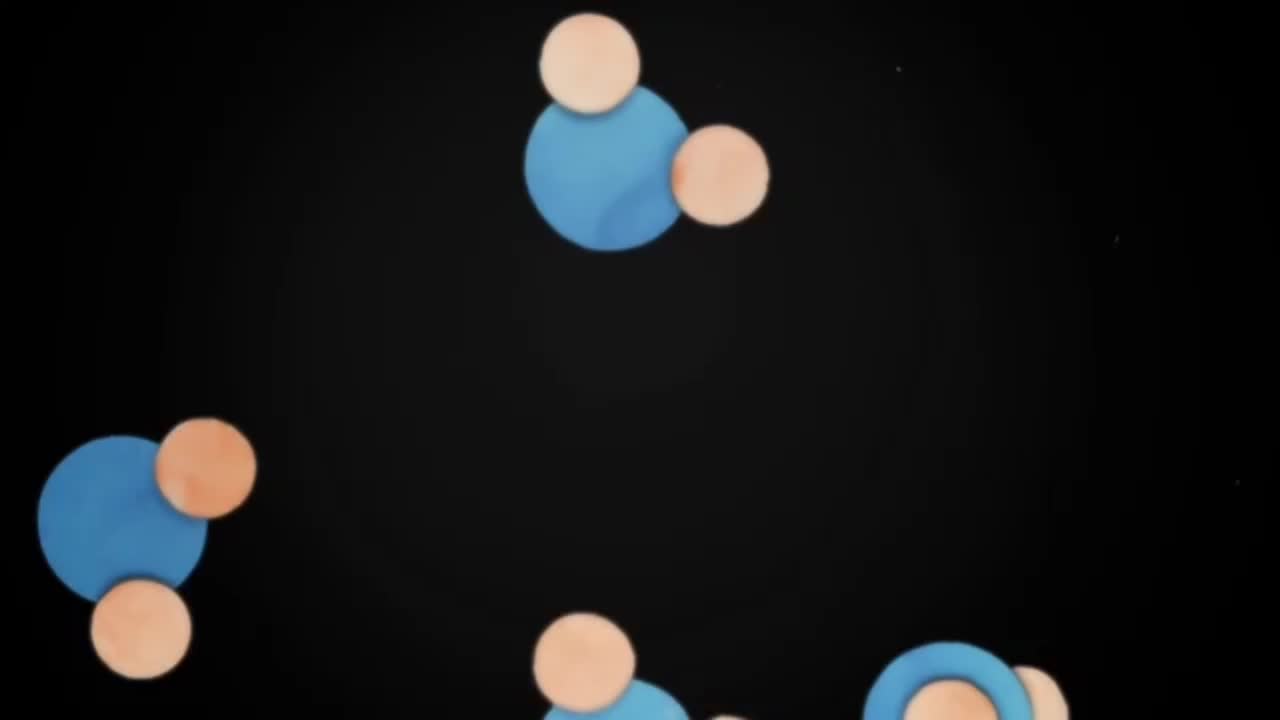
How do hydrogen bonds form
3 years ago
3.07K
The water molecule as a whole is electrically neutral,but oxygen gets a larger share of electrons,making it slightly negative and the hydrogens slightly positive.Due to its negative charge,the oxygen in one molecule is attracted to the positive charge of the hydrogen in another molecule.And so a weak bond between the two molecules,called a hydrogen bond,is formed.
Loading comments...
-
 LIVE
LIVE
LFA TV
13 hours agoLFA TV ALL DAY STREAM - THURSDAY 8/28/25
822 watching -
 LIVE
LIVE
LIVE WITH CHRIS'WORLD
4 hours agoLIVE WITH CHRIS’WORLD - YOU DON’T HATE THE MEDIA ENOUGH!
151 watching -
 LIVE
LIVE
SavageJayGatsby
1 day agoFirst Rumble Exclusive Stream?! | Let's Play: Prey | $300 Weekly Goal for Spicy Bite Saturday
161 watching -
 11:42:24
11:42:24
GritsGG
12 hours agoWin Streaking! Most Wins 3485+ 🧠
36.9K1 -
 LIVE
LIVE
Quite Frankly
6 hours ago"Mixed News, RFK Pull-up Challenge, Calls" ft. J Gulinello 8/28/25
525 watching -
 1:14:52
1:14:52
TheCrucible
4 hours agoThe Extravaganza! EP: 29 (8/28/25)
93.8K8 -
 1:09:58
1:09:58
Kim Iversen
4 hours agoTrans. Russian. Anti-Israel. Anti-Trump. Are You Buying This Story?
41.8K77 -
 1:51:08
1:51:08
Redacted News
4 hours agoEMERGENCY! BILL GATES CULT MEMBERS FOUND PLANTED INSIDE MULTIPLE FEDERAL AGENCIES, RFK FURIOUS
140K110 -
 31:02
31:02
Kimberly Guilfoyle
5 hours agoFull Breaking News Coverage: Live with John Nantz & Steve Moore | Ep250
28.6K13 -
 1:15:19
1:15:19
vivafrei
6 hours agoShameless Politicization of Tragedy! Susan Monarez is OUT! Pritzker is an IDIOT! & MORE!
130K53