Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
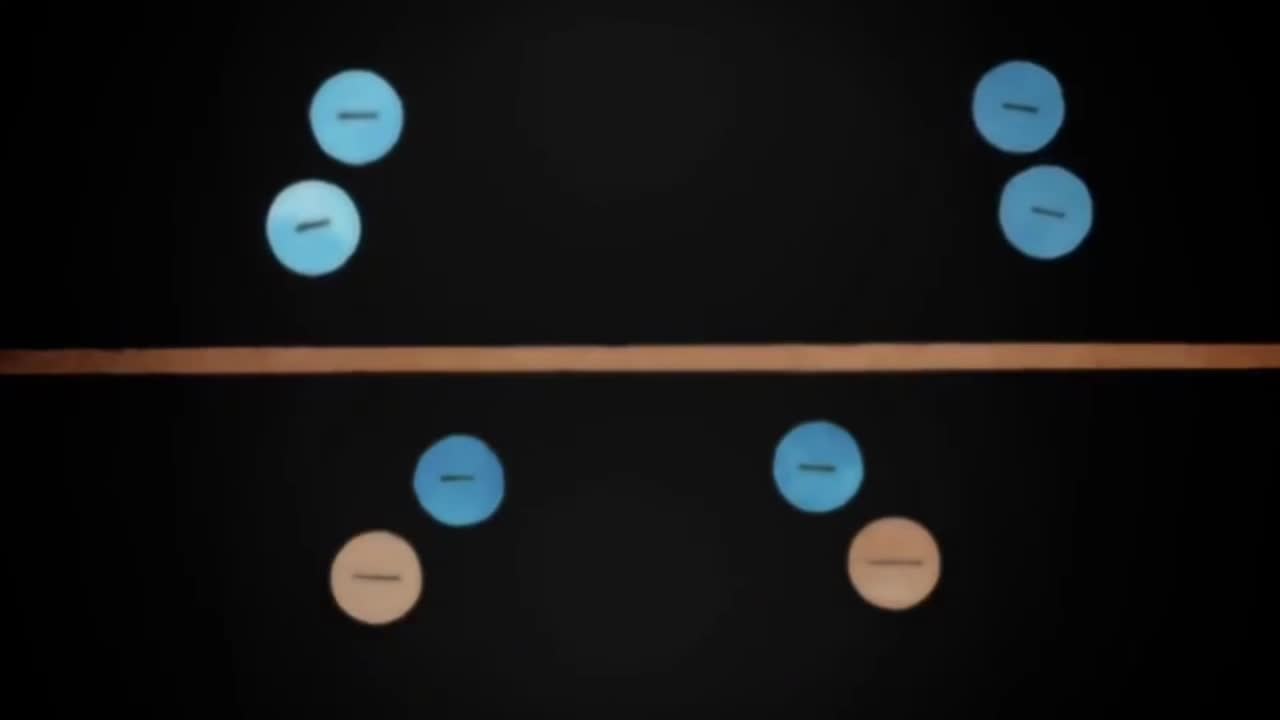
The relationship between electron charge and hydrogen atom
3 years ago
3.03K
Similar charges repel,so they tend to stay as far away from each other as possible.The pairs form four electron clouds,two of which are where the hydrogen and oxygen share electrons.The repulsion between the unbonded pairs is even stronger than repulsion between the shared pairs,so the two hydrogens get pushed a little further to an angle of 104.5 degrees.
Loading comments...
-
 LIVE
LIVE
SpartakusLIVE
57 minutes ago#1 Saturday Spartoons || HUGE Announcement TODAY
432 watching -
 8:31
8:31
MattMorseTV
2 hours ago $0.06 earnedTexas just did the IMPOSSIBLE.
4.07K45 -
 24:39
24:39
MYLUNCHBREAK CHANNEL PAGE
1 day agoInterdimensional Beings at Borobudur
17.4K11 -
 12:42
12:42
Scammer Payback
21 hours agoCalling Scammers who were Raided
1.65K6 -
 23:31
23:31
IsaacButterfield
13 hours agoThe Woke Mob Is Really CANCELLING Matt Rife For THIS…
9324 -
 1:23
1:23
WildCreatures
8 days ago $0.90 earnedThis mother armadillo eating her palm nuts is truly adorable
1.83K9 -
 8:59
8:59
The Art of Improvement
8 hours agoHow to Build the Most Powerful Mindset for Success
68 -
 LIVE
LIVE
GritsGG
1 day ago36 Hour Stream! Most Wins 3420+ 🧠
1,224 watching -
 1:26:16
1:26:16
Michael Franzese
18 hours agoMenendez Brothers Denied Parole – Newsom Holds Their Fate
105K79 -
 2:36:02
2:36:02
I_Came_With_Fire_Podcast
16 hours agoSecret Origins of Transhumanism & The New Atlantis
28.2K16