Premium Only Content
This video is only available to Rumble Premium subscribers. Subscribe to
enjoy exclusive content and ad-free viewing.
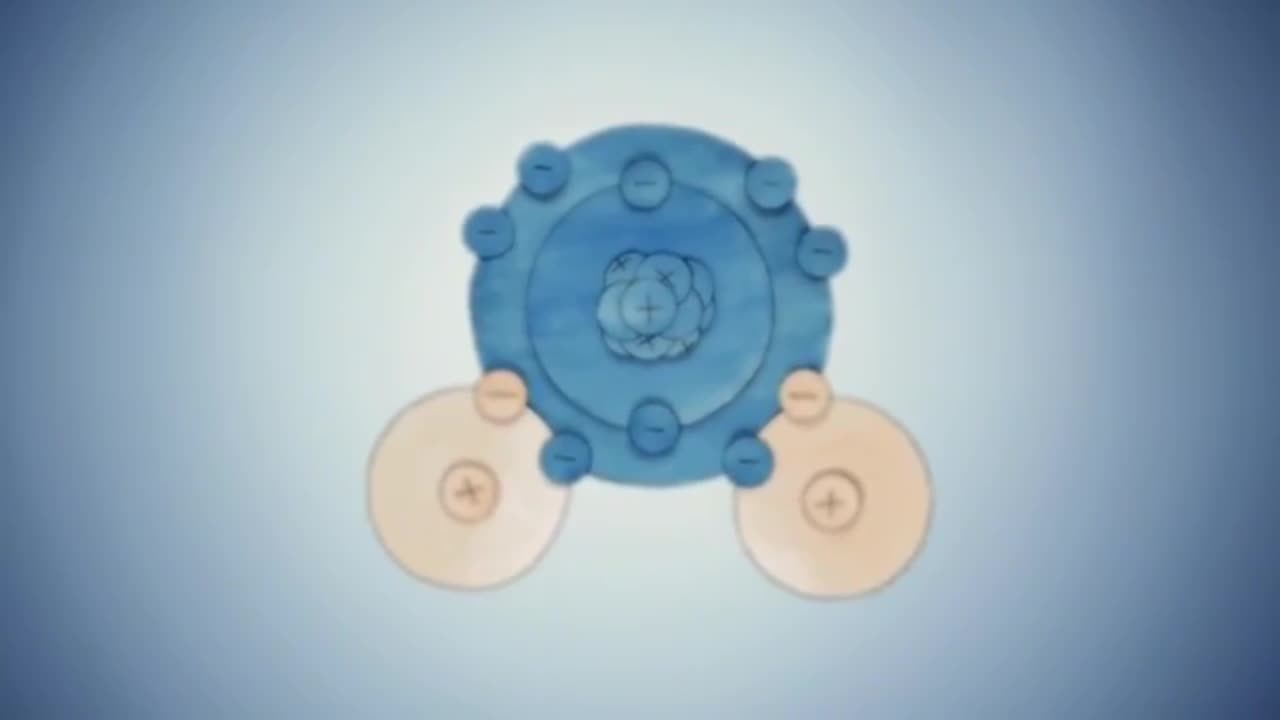
The principle between electrons in the composition of water
3 years ago
3.04K
The two electrons from oxygen's outer shell are shared with two electrons from both hydrogens as they bond together,and the remaining four outer shell electrons from oxygen form two pairs.We call the bonds between these atoms covalent bonds.The pairs of electrons are all negatively charged.
Loading comments...
-
 LIVE
LIVE
Lofi Girl
2 years agoSynthwave Radio 🌌 - beats to chill/game to
525 watching -
 5:52:00
5:52:00
Akademiks
12 hours agoWAR IN ATLANTA Episode 5. YOUNG THUG FIRST INTERVIEW about SNITCHING, GUNNA... BREAKS DOWN CRYING!
165K9 -
 7:30:25
7:30:25
SpartakusLIVE
15 hours agoVerdansk Duos w/ Nicky || Saturday Spartoons - Variety Later?!
62.3K1 -
 1:38:47
1:38:47
Badlands Media
1 day agoDevolution Power Hour Ep. 387: Trump, Epstein, Durham Mysteries, and North Korea Ops
104K27 -
 1:05:23
1:05:23
Man in America
19 hours agoSoaring Gold Exposes the Imminent Crash of the Old System w/ John Perez
61.6K21 -
 2:42:40
2:42:40
TruthStream with Joe and Scott
19 hours agoTHOMAS AND GROK: AI, Bible decodes, The JESUS Cube live 9/6 #487
50.7K11 -
 2:34:46
2:34:46
BlackDiamondGunsandGear
14 hours agoGet Prepped / After Hours Armory / LIVE SHOW /
33.1K2 -
 2:01:39
2:01:39
Tundra Tactical
12 hours ago $11.29 earned🛑LIVE NOW!! This spits in the face of the Second Amendment.🛑
39.9K8 -
 2:34:46
2:34:46
DLDAfterDark
11 hours ago $4.97 earnedIt's SHTF! Do You Have What You Need?? Let's Review Items & Priorities
28.2K6 -
 28:58
28:58
Stephen Gardner
12 hours ago🚨Explosive allegations: Rosie O’Donnell connects Trump to Epstein scandal!?
45.5K86